What are the characteristics of an exothermic reaction
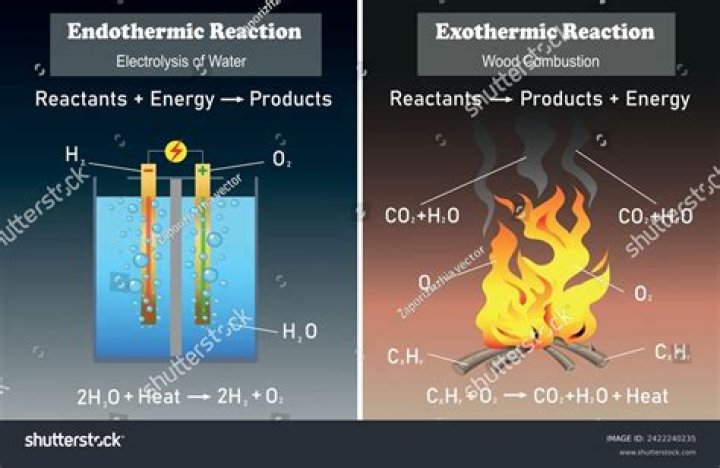
Exothermic reactions are reactions or processes that release energy, usually in the form of heat or light. In an exothermic reaction, energy is released because the total energy of the products is less than the total energy of the reactants.
What are 3 exothermic reactions?
- any combustion reaction.
- a neutralization reaction.
- rusting of iron (rust steel wool with vinegar)
- the thermite reaction.
- reaction between water and calcium chloride.
- reaction between sodium sulfite and bleach (dilute sodium hypochlorite)
- reaction between potassium permanganate and glycerol.
What is the defining characteristic of an endothermic reaction?
A chemical reaction will either absorb energy from its surroundings or release energy into its surroundings. A reaction that absorbs energy is said to be endothermic. In an endothermic reaction, the starting materials (reactants) are more stable than the products; they are in a lower energy state.
What are the characteristics of an exothermic and an endothermic reaction?
An exothermic process releases heat, causing the temperature of the immediate surroundings to rise. An endothermic process absorbs heat and cools the surroundings.”What are 5 exothermic reactions?
- Making of an ice cube. Making an ice cube is a process of liquid changing its state to solid. …
- Snow formation in clouds. …
- Burning of a candle. …
- Rusting of iron. …
- Burning of sugar. …
- Formation of ion pairs. …
- Reaction of Strong acid and Water. …
- Water and calcium chloride.
How do you determine if a reaction is exothermic?
The general equation for an exothermic reaction is: Reactants → Products + Energy. Note: ΔH represents the change in energy. If the energy produced in an exothermic reaction is released as heat, it results in a rise in temperature.
What are 5 examples of exothermic reactions?
Brushing your teeth, washing your hair, and lighting your stove are all examples of exothermic reactions. Keep reading to learn about combustion, neutralization, corrosion, and water-based exothermic reactions.
What defines exothermic reaction?
In thermochemistry, an exothermic reaction is a “reaction for which the overall standard enthalpy change ΔH⚬ is negative.” Exothermic reactions usually release heat and entail the replacement of weak bonds with stronger ones.Which answer defines exothermic reaction?
An exothermic reaction has reactants that are lower in energy than products because energy is released to form the products. An exothermic reaction has reactants that are higher in energy than products because energy is absorbed to form the products.
What are the characteristics of a reaction?- Evolution of gas.
- Formation of a precipitate.
- Change in color.
- Change in temperature.
- Change in state.
- Formation of new substances.
What are the characteristics of a combustion reaction?
Combustion means burning, usually in oxygen but sometimes with other oxidants such as fluorine. A combustion reaction happens quickly, producing heat, and usually light and fire. Combustion describes how the reaction happens, not the reactants and products.
Which is not a characteristic of endothermic reaction?
Explanation: An endothermic reaction absorbs energy in the form of heat. … Since endothermic reactions involve absorbing energy, often in the form of heat, the change in enthalpy is positive. Therefore, the answer is “releasing of energy to surroundings” which is not a characteristic of an endothermic process.
What is an exothermic reaction give an example Class 7?
A reaction that is chemical in nature and is characterized by the release of energy in the form of heat or light is called an exothermic reaction. Matching a light using a matchstick is one example of this type of reaction where the release is in the form of both heat and light.
What are the 5 characteristics of a chemical reaction?
The five conditions of chemical change: color change, formation of a precipitate, formation of a gas, odor change, temperature change.
What are the 5 characteristics of a chemical reaction with examples?
The characteristics of a chemical reaction are: Evolution of gas, Formation of a precipitate, Change in colour, Change in temperature, Change in state etc.
Why are combustion reaction exothermic?
Combustion is an oxidation reaction that produces heat, and it is therefore always exothermic. All chemical reactions first break bonds and then make new ones to form new materials. … If the energy released by the new bonds is greater than the energy needed to break the original bonds, the reaction is exothermic.
What are the characteristics of a flame?
- Flame height is measured vertically from ground level to the flame tip. …
- Fire intensity is the rate at which energy (heat) is released as the fire burns. …
- Season of year affects the impact of a burn.
What is an exothermic reaction Class 8?
An exothermic reaction is a chemical reaction in which less energy is needed to break bonds in the reactants than is released when new bonds form in the products. During an exothermic reaction, energy is constantly given off, often in the form of heat. All combustion reactions are exothermic reactions.
What is exothermic reaction for class 10th?
A chemical reaction in which a large amount of heat / energy is produced along with the products is called exothermic reaction.
What is exothermic reaction give two examples Class 10?
Exothermic reactions: The chemical reaction in which heat is given out is called exothermic reactions. It causes rise in temperature. . e.g. (i) When carbon bums in oxygen to form carbon dioxide, a lot of heat is produced. C + O2 → CO2 + heat.
What are characteristics Class 10?
Hint: Characteristics are often referred to as features, these are the unique properties of an individual or any substance or topic which can be used to define it and makes it unique.
What are the 7 signs of a chemical reaction?
- Gas Bubbles Appear. Gas bubbles appear after a chemical reaction has occurred and the mixture becomes saturated with gas. …
- Formation of a Precipitate. …
- Color Change. …
- Temperature Change. …
- Production of Light. …
- Volume Change. …
- Change in Smell or Taste.
What are the characteristics of chemical reactions Class 10?
- 1)Evolution of gas. Some chemical reactions are characterised by the evolution of gas. …
- 2)Formation of precipitate. Some chemical reactions are characterised by the formation of precipitate. …
- 3)Change in colour. …
- 4)Change in temperature. …
- 5)Change of state.



