Why is high salinity bad
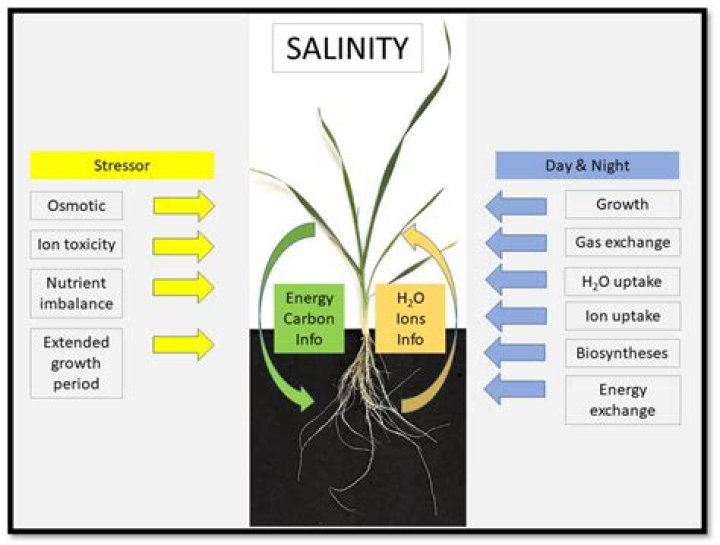
If the level of salts in the soil water is too high, water may flow from the plant roots back into the soil. … Salinity affects production in crops, pastures and trees by interfering with nitrogen uptake, reducing growth and stopping plant reproduction.
Why is increased salinity bad?
Salt stressed soils are known to suppress the growth of plants (Paul, 2012). … Crops grown on saline soils suffer on an account of high osmotic stress, nutritional disorders and toxicities, poor soil physical conditions and reduced crop productivity.
What are the consequences of salinity?
Salinity affects: farms – salinity can decrease plant growth and water quality resulting in lower crop yields and degraded stock water supplies. Excess salt affects overall soil health, reducing productivity. It kills plants, leaving bare soil that is prone to erosion.
How salinity affect water quality?
Salinity affects water density. The higher the dissolved salt concentration, the higher the density of water 4. The increase in density with salt levels is one of the driving forces behind ocean circulation 22.Why is salinity a problem in Australia?
Salinity has been caused by extensive land clearing in Australia, predominantly for agricultural purposes. … Land clearance can also lead to soil erosion and, when it results in a changing water balance, to dryland salinity.
How does salinity affect density?
Salinity Affects Density When salt is dissolved in fresh water, the density of the water increases because the mass of the water increases. … When comparing two samples of water with the same volume, the water sample with higher salinity will have greater mass, and it will therefore be more dense.
What is high salinity?
The concentration of dissolved salt in a given volume of water is called salinity. Salinity is either expressed in grams of salt per kilogram of water, or in parts per thousand (ppt, or ‰). … Water with salinity above 50 ppt is brine water, though not many organisms can survive in such a high salt concentration.
How does salinity affect species diversity?
Results confirm salinity as a strong determinant of phytoplankton diversity, while also demonstrating the importance of nutrient supply, where species richness decreased with increasing salinity and increased with nutrient enrichment.What is high salinity in water?
Slightly saline water – From 1,000 ppm to 3,000 ppm. Moderately saline water – From 3,000 ppm to 10,000 ppm. Highly saline water – From 10,000 ppm to 35,000 ppm.
How does salinity affect animals?Salinities at the margins or outside the tolerance range of particular species will prevent their occurrence, change their behaviour, or limit reproduction and germination, reducing their fitness for survival in that environment.
Article first time published onHow does salt affect the environment?
At high concentrations, salt can be fatal to some aquatic animals. Salt can also change the way the water mixes and lead to the formation of salty pockets near the bottom of lakes, creating biological dead zones. … In fact, almost all chloride ions from the road salt eventually find their way into waterways downstream.
Is salinity a big problem in Australia?
Excessive amounts of dissolved salt in water can affect agriculture, drinking water supplies and ecosystem health. Salinity is a significant issue in south-western Australia and in some Murray-Darling Basin regions within New South Wales, Victoria, and South Australia.
How much land is affected by salinity in Australia?
Dryland salinity affects almost 2.5 million hectares of Australian farm land, and is expanding at a rate of 3-5 per cent a year, at an estimated annual cost of $270 million.
How does soil salinity affect the environment?
The main point is that excess salinity in soil water can decrease plant available water and cause plant stress. … As the water is taken up by plants through transpiration or lost to the atmosphere by evaporation, soil water salinity increases because salts become more concentrated in the remaining soil water.
What is salinity and why does it matter?
Salinity is the dissolved salt content of a body of water. It is a strong contributor to conductivity and helps determine many aspects of the chemistry of natural waters and the biological processes within them.
What are the reasons of differences in salinity on a global level?
Evaporation of ocean water and formation of sea ice both increase the salinity of the ocean. However these “salinity raising” factors are continually counterbalanced by processes that decrease salinity such as the continuous input of fresh water from rivers, precipitation of rain and snow, and melting of ice.
Is salinity abiotic or biotic?
Salinity is an important abiotic factor because the normal functioning of animals depends on the regulation of the water and ions in their internal environment, which is influenced by the water and ions in their external environment (Moyes & Schulte 2006).
Why do changes in salinity affect living organisms?
Why Salinity Is Important Salinity can affect the density of ocean water: Water that has higher salinity is denser and heavier and will sink underneath less saline, warmer water. This can affect the movement of ocean currents. It can also affect marine life, which may need to regulate its intake of saltwater.
Is salinity higher in warm or cold water?
Salinity, Density, and Temperature Extra space is created by this expansion into which salt and other molecules (e.g., calcium) can fit. Since warmer water thus can hold more salt and other molecules than cold water; it can have a higher salinity.
How does salinity affect buoyancy?
How Salt Water Influences Buoyancy. Adding salt increases the density of water. … If the same volume of water is displaced by an object, the weight of saltwater displaced is greater and thus the force of buoyancy is proportionally greater.
How does salinity affect freshwater ecosystems?
In the summer, estuaries increase in salinity with decreased freshwater inflows and increased evaporation due to higher temperatures causing the system to be classified as a negative estuarine system. Salinity affects the chemical conditions within an estuary, most notably the amount of dissolved oxygen.
How does salinity affect dissolved oxygen?
How does salinity affect Dissolved Oxygen solubility? Dissolved oxygen decreases exponentially as salt levels increase. That is why, at the same pressure and temperature, saltwater holds about 20% less dissolved oxygen than freshwater.
How salinity is caused?
Primary salinity is caused by natural processes such the accumulation of salt from rainfall over many thousands of years or from the weathering of rocks.
Does salinity affect primary production?
Therefore the study concluded that salinity had a clear effect on the primary productivity, and the study suggested that salinity, due to climate change, could be the main key driver for primary productivity in the subtropics but still needs more deeply field studies in different subtropical regions.
How does salinity affect zooplankton?
Salinity negatively affected the abundances of rotifers, cladocerans, and total zooplankton, while it had little effect on copepod abundance. … In general, salinity was the most important environmental factor shaping zooplankton biodiversity and abundance.
What is the relationship between salinity nutrients and density?
Salinity, temperature and depth all affect the density of seawater. Temperature and salinity affect water density. Cold water is denser than warm water. Freshwater is less dense than seawater.
What happens if there is too much salt in the ocean?
A few hours later, virtually all marine life would die as their cells swelled and ruptured due to osmosis (water molecules move towards areas of higher salt concentration). They would sink to the ocean floor, but their bodies wouldn’t decompose, because all marine bacteria would be dead too.
Why is global warming affecting the salinity of the world's oceans and why is this important what effects does this have?
Changes in salinity could also affect water currents because saltwater is denser than fresh water and sinks. Warmer air can absorb more water than cooler air, so as the climate warms, more water can evaporate into the air. … That intensifies the water cycle on both ends of the spectrum.
Is mgso4 bad for the environment?
Magnesium can be toxic at concentrations approaching natural background levels, but toxicity is dependent on Ca concentrations, with exposure in very low ionic concentration, Ca-deficient waters posing the greatest risk to aquatic life.
Is table salt bad for the environment?
Sodium chloride is not classified as dangerous to the environment and the manufacture of salt does not require registration under the Environmental Protection Act Integrated Pollution and Control Regulations.
Why is the salt for snow bad for the environment?
As snow and ice melts, the sodium chloride runs off into fields and streams, which can make them uninhabitable. Over time, the buildup of road salt can be dangerous to vegetation, wildlife and fish. The buildup can also be dangerous if it contaminates a city’s water supply.