Why is a salt bridge used in chemistry?
Why is a salt bridge used in chemistry?
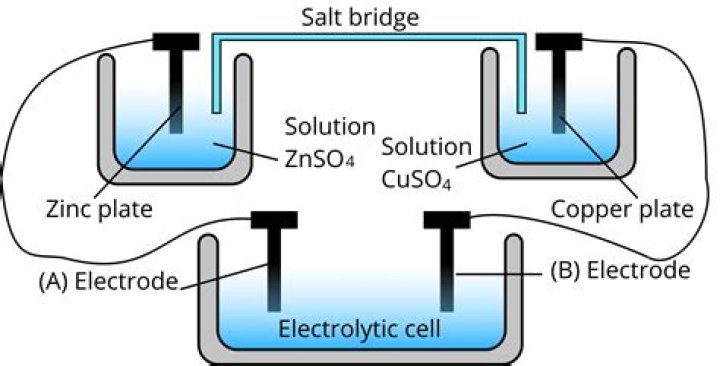
Why a Salt Bridge Is in Chemistry and Why One Is Used. A salt bridge is a connection containing a weak electrolyte between the oxidation and reduction half-cells in a galvanic cell (e.g., voltaic cell, Daniell cell). Its purpose is to keep the electrochemical reaction from reaching equilibrium too quickly.
How is the free energy of the salt bridge calculated?
Titration curve between the wild-type (blue) and the mutant (red) The second method utilizes nuclear magnetic resonance spectroscopy to calculate the free energy of the salt bridge. A titration is performed, while recording the chemical shift corresponding to the protons of the carbon adjacent to the carboxylate or ammonium group.
What happens if a cell is constructed without a salt bridge?
If a cell is constructed without a salt bridge, one solution would quickly accumulate positive charge while the other would accumulate negative charge. This would halt the reaction and thus the generation of electricity. The two main types of salt bridges are a glass tube and a piece of filter paper:
Which is a non-covalent interaction in a salt bridge?
Salt bridge (protein and supramolecular) In chemistry, a salt bridge is a combination of two non-covalent interactions: hydrogen bonding and ionic bonding (Figure 1). Ion pairing is one of the most important noncovalent forces in chemistry, in biological systems, in different materials and in many applications such as ion pair chromatography.
How to prepare an electrochemical salt bridge for use?
Let’s go through how to prepare an electrochemical salt bridge. It is a fairly simple device and can just be a string, or a piece of cotton that has been soaked in an aqueous solution like sodium sulfate, for instance. If you choose to make a salt bridge out of string or cotton, it shouldn’t be soaking wet with solution.
What is the function of a salt bridge?
Salt Bridge Function. The main function of a salt bridge is to help maintain the electrical neutrality within the internal circuit. It also helps in preventing the cell from taking its reaction to equilibrium. If salt bridges are absent or if they are not used then the reaction will likely continue and the solution in one-half electrodes will
What makes a salt bridge in a brine tank?
A salt bridge is a situation in which the salt forms a surface crust spanning the water in the brine tank. Viewed from above, it may look like a normal layer of salt pellets, but this crust is on the surface only—below it is open space.
What do you need to make an Agar Salt Bridge?
It typically comes in powered form. To make an agar salt bridge, around 2-5% agar is mixed into a 1 molar solution of sodium sulfate. You only need around 3 mL to make enough material for 50 salt bridges. This mixture should be heated slowly while stirring until it is turns clear and bubbles just start to be seen.