Why does Hesss law work
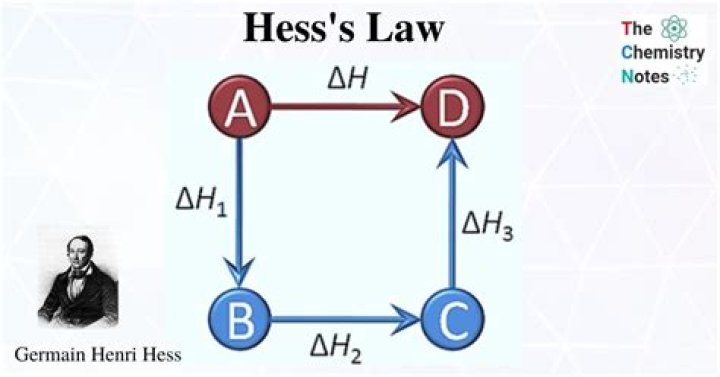
Hess’s law is due to enthalpy being a state function, which allows us to calculate the overall change in enthalpy by simply summing up the changes for each step of the way, until product is formed. All steps have to proceed at the same temperature and the equations for the individual steps must balance out.
Why is Hess's law valid?
If a process written as the sum of several stepwise processes, the enthalpy change of the total process equals the sum of the enthalpy changes of the various steps. Hess’s law is valid because enthalpy is a state function.
How do I verify Hess law?
Finally, to verify Hess’s Law, you will use the H’s you measured for the first two reactions to calculate H for the third reaction. Compare it to what you measured for the third reaction by calculating the Percent Error.
How does Hess's law allow you to determine the enthalpy of a reaction without carrying out the reaction?
Hess’s law allows us to calculate ΔH values for reactions that are difficult to carry out directly by adding together the known ΔH values for individual steps that give the overall reaction, even though the overall reaction may not actually occur via those steps.What is thermodynamic basis of Hess's law?
Hess’s law states that the energy change in an overall chemical reaction is equal to the sum of the energy changes in the individual reactions comprising it. … The law is a variation of the first law of thermodynamics and conservation of energy.
Can you add in Hess law?
To use Hess’s law, we need to determine how the three equations above can be manipulated so that they can be added together to result in the desired equation (the formation of acetylene from carbon and hydrogen). … When one mole of acetylene is produced, 228.3 kJ of heat are absorbed, making the reaction endothermic.
What data is needed to use Hess's law?
In order to use Hess’ Law one would have to utilize data such as enthalpy changes and the individual thermochemical equations.
What does Hess's law require us to do to the ΔH of a thermochemical equation if we reverse the equation?
Hess’s law states that when chemical equations are combined algebraically, their enthalpies can be combined in exactly the same way. … If a chemical reaction is reversed, the sign on ΔH is changed. If a multiple of a chemical reaction is taken, the same multiple of the ΔH is taken as well.What is Hess law example?
Hess’s Law Example. Experiments were performed to determine the heat of combustion of methanol, formaldehyde, and hydrogen. Using this data determine the DHrxn for the conversion of methanol to formaldehyde and hydrogen. Rearrange the equations so that they add up to the reaction of interest.
What is coffee cup calorimetry?A coffee cup calorimeter is used to measure enthalpy changes in chemical processes, giving ΔH. Essentially, the heat measured in the device is equivalent to ΔH, the change in enthalpy.
Article first time published onCan Hess's law be proved experimentally?
(3) Heat of formation of an atom =1//2xx bond energy like atoms covalent bond. (4) The heat enthalpy change in a chemical reaction is equal but opposite to the heat enthalpy if reaction is reversed. (5) Hess’s law can be verified experimentally. Hess’s law can not be verified experimentally.
What is Hess law class 11?
The Hess’s law states that the total enthalpy change during a complete chemical reaction is the same regardless of the path taken by the chemical reaction. … Hess’s law can be seen as an application of the principle of conservation of energy.
What is the importance of Hess law of constant heat summation?
Hess’s law of heat summation is of great importance as it chemically represents the laws of thermodynamics and the conservation of energy.
What is the most important application of Hess's law?
Marta O. The most famous applications of the Hess law are reactions which happen in our body when we consume food and in the industry to see how much energy the engine produces/ uses.
What is Hess's law and its application?
Applications of Hess’s Law: Thermochemical equations can be added subtracted or multiplied like ordinary algebraic equations. Hess’s law is useful to calculate heats of many reactions which do not take place directly. … It is useful to find out the heat of formation, neutralization, etc.
Which substance has defined as 0 kJ mol?
CompoundΔHfoH2(g)0 kJ/molH2O(g)-241.8 kJ/molHF(g)-271.1 kJ/molNO(g)90.25 kJ/mol
Can you divide Hess law?
Hess’s law states that the standard reaction enthalpy is the sum of the standard enthalpies of the intermediate reactions into which the overall reaction can be divided, while each occurs at the same temperature.
What does Hess law say about the enthalpy of a reaction apex?
The law states that the total enthalpy change during a reaction is the same whether the reaction is made in one step or in several steps. … Hess’ law allows the enthalpy change (ΔH) for a reaction to be calculated even when it cannot be measured directly.
What is Hess law of constant heat summation explain with example?
Hess’s law, also called Hess’s law of constant heat summation or Hess’s law of heat summation, rule first enunciated by Germain Henri Hess, a Swiss-born Russian chemist, in 1840, stating that the heat absorbed or evolved (or the change in enthalpy) in any chemical reaction is a fixed quantity and is independent of the …
Why do you put a lid on the Styrofoam cup?
Coffee cups, especially those made of Styrofoam, are effective calorimeters because they hold in the heat. … The lid should be flat and make a good seal when placed inside the lip of the cup. Cardboard acts as a better insulator than plastic lids do.
What is bomb calorimetry?
Bomb calorimeters A bomb calorimeter is a type of constant-volume calorimeter used in measuring the heat of combustion of a particular reaction. Bomb calorimeters have to withstand the large pressure within the calorimeter as the reaction is being measured.
Why are bomb calorimeters better?
IN CONCLUSION. In conclusion, the bomb calorimeter is more accurate and leaves less room for human error. Our bomb calorimeters use a digital temperature sensor with a 22 bit resolution versus an analogue glass tube thermometer used in a coffee cup calorimeter.
Why should you not look at burning magnesium?
The burning magnesium ribbon produces light of sufficient intensity to cause temporary loss of sight. Avoid looking directly at the light source. The burning of magnesium in air produces intense heat which can cause burns and initiate combustion in flammable materials.
How does Hess's law compare to the law of conservation of energy?
The Hess’s law states that the standard enthalpy of an overall reaction is the summation of the enthalpy changes in the individual reactions. The law of conservation of energy states that the total energy of an isolated system remains constant.
How does Hess's law relate to calorimetry?
Since it is difficult to measure the enthalpy of combustion of a metal directly, in this lab it will be determined indirectly by applying Hess’s Law of Heat Summation. Hess’s Law states that the enthalpy change of an overall process is equal to the sum of the enthalpy changes of its individual steps.
What is Hess law BYJU's?
Hess’ law also known as Hess’s law of constant heat summation states, “at constant temperature, heat energy changes (enthalpy – ΔHrec) accompanying a chemical reaction will remain constant, irrespective of the way the reactants react to form product”.
Which chapter is Hess law?
Chapter 15.4: Hess’s Law.
What is the Hess's law write its mathematical expression?
In mathematical form; Consider , the enthalpy of an overall reaction i.e. A to B along one path is ΔHand ΔH1, ΔH2, ΔH3 and ΔH4 representing enthalpies of reactions leading to the same product B, along another path, then; ΔH=ΔH1+ΔH2+ΔH3+ΔH4.
What is Hess's law of heat summation give its two important applications?
Hess law of constant heat summation states that the total enthalpy change during a reaction is the same whether the reaction takes place in one step or in several steps. … Application: Hess law is useful to calculate heats of many reactions which do not take place directly.