Why can boiling points and freezing points be used to identify substances
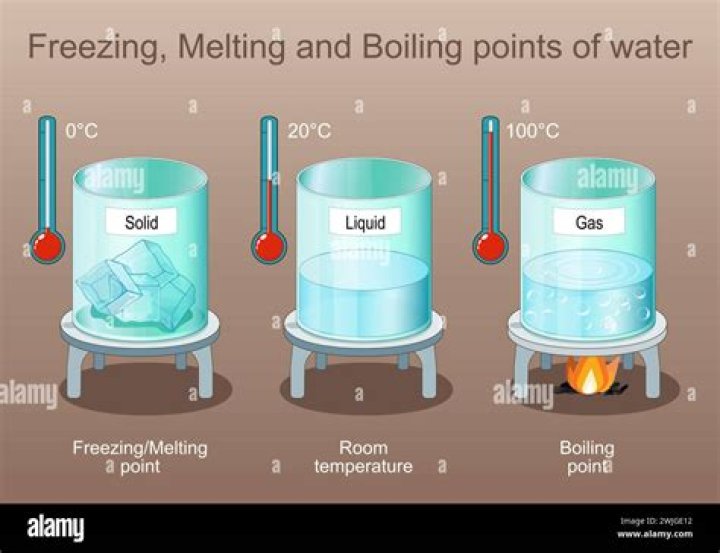
Scientists determine melting or boiling point by heating a small sample of the substance, keeping careful track of the temperature the entire time. When they see a solid melt, they record the temperature as the melting point. When they see a liquid boil, they record the temperature as the boiling point.
How can boiling point be used to identify a substance?
Scientists determine melting or boiling point by heating a small sample of the substance, keeping careful track of the temperature the entire time. When they see a solid melt, they record the temperature as the melting point. When they see a liquid boil, they record the temperature as the boiling point.
What properties can be used to identify substances?
These properties include color, odor, taste, density, melting point, boiling point, conductivity, and hardness. Chemical properties tell us how the substance interacts with other substances and may include reaction with oxygen (oxidation), chlorine, metals, etc..
Why is melting point a property that can be used to identify a substance?
The melting point is a physical property of a solid and can be used to help identify a substance. In practice, a solid usually melts over a range of temperatures rather than at one specific temperature. For this reason it is more useful to speak of a melting point range.What is the significance of boiling point and melting point of a substance?
The boiling point is the temperature at which a material changes from a liquid to a gas (boils) while the melting point is the temperature at which a material changes from a solid to a liquid (melts).
Why is boiling point considered a physical property?
When something is boiling, its phase changes from the liquid phase to the gas phase; no chemical bonds are broken; molecules become farther removed from each other; and the transition qualifies as a physical change.
Why are boiling points important?
The boiling point of organic compounds can give important information about their physical properties and structural characteristics. Boiling point helps identify and characterise a compound. … A liquid at a higher pressure has a higher boiling point than when that liquid is at lower atmospheric pressure.
Why compound should not be heated to the point that it melts?
If you keep making the molecules bigger, at some point, the melting temperature would be higher than the decomposition temperature. If you heat such compounds, they decompose before they can melt.Why is boiling point considered a physical property when a gas appearance is much different from that of a liquid?
They are also physical changes because they do not change the nature of the substance. Boiling water: Boiling water is an example of a physical change and not a chemical change because the water vapor still has the same molecular structure as liquid water (H2O).
Is boiling point a physical or chemical properties?Characteristics such as melting point, boiling point, density, solubility, color, odor, etc. are physical properties. Properties that describe how a substance changes identity to produce a new substance are chemical properties.
Article first time published onWhat is the best way to identify a substance?
Physical properties of a substance are characteristics that can be observed without altering the identity of the substance. Color, odor, density, melting temperature, boiling temperature, and solubility are examples of physical properties. Physical properties can be used to identify a pure substance.
How do you identify substances?
- chemical name, for example, benzene;
- number, for example, EC number 200-753-7, and.
- chemical composition, for example, >99 % benzene and <1 % toluene. The composition is determined by chemical analysis.
Why are melting point and boiling point different for the same matter?
This is when they become a liquid. So the melting point is the temperature at which molecules in a solid can move past each other and form a liquid. The boiling point, on the other hand, involves liquids and gases. … As temperature is increased, the liquid molecules move faster and faster and more of them escape as gas.
What is the effect of pressure on boiling point and melting point?
Both the melting and boiling point increase with increase in pressure and decrease with decrease in pressure as it is directly proportional to the pressure.
What does boiling point mean in chemistry?
boiling point, temperature at which the pressure exerted by the surroundings upon a liquid is equaled by the pressure exerted by the vapour of the liquid; under this condition, addition of heat results in the transformation of the liquid into its vapour without raising the temperature.
What happens when a substance reaches its boiling point?
When a liquid is heated, it eventually reaches a temperature at which the vapor pressure is large enough that bubbles form inside the body of the liquid. This temperature is called the boiling point. Once the liquid starts to boil, the temperature remains constant until all of the liquid has been converted to a gas.
What affects the boiling point of a substance?
The boiling point of a liquid depends on temperature, atmospheric pressure, and the vapor pressure of the liquid.
Is the boiling point of a substance considered a physical property Why or why not?
A physical property is a characteristic of matter that is not associated with a change in its chemical composition. Familiar examples of physical properties include density, color, hardness, melting and boiling points, and electrical conductivity.
What is the difference between the melting point and freezing point of a substance?
Freezing point is the temperature at which a liquid becomes a solid at normal atmospheric pressure. Alternatively, a melting point is the temperature at which a solid becomes a liquid at normal atmospheric pressure.
What are the properties of boiling point?
Boiling point is the temperature at which the vapor pressure of a chemical equals atmospheric pressure. To simply put, it measures the temperature at which a chemical boils. Similar to melting point, a higher boiling point indicates greater inter-molecular forces and therefore less vapour pressure.
Are temperature weight boiling point melting point color and density physical or chemical properties?
A physical property is a characteristic of matter that is not associated with a change in its chemical composition. Familiar examples of physical properties include density, color, hardness, melting and boiling points, and electrical conductivity.
How can we utilize properties like boiling point and melting point in testing the purity of a substance?
Melting and Boiling Point Determination The physical properties of a substance can be used to establish its purity. These properties include the melting point and boiling point. Different substances tend to have different melting and boiling points, and any pure substance will have a specific melting and boiling point.
Why is hardness a physical property?
Physical properties are those that can be observed without changing the identity of the substance. The general properties of matter such as color, density, hardness, are examples of physical properties. … Flammability and corrosion/oxidation resistance are examples of chemical properties.
What is freezing and freezing point?
freezing point, temperature at which a liquid becomes a solid. As with the melting point, increased pressure usually raises the freezing point. The freezing point is lower than the melting point in the case of mixtures and for certain organic compounds such as fats.
For what two purposes are melting points routinely used?
Melting points are often used to characterize organic and inorganic crystalline compounds and to ascertain their purity.
What affects the melting point of a compound?
The force of attraction between the molecules affects the melting point of a compound. Stronger intermolecular interactions result in higher melting points. … In organic compounds the presence of polarity, or especially hydrogen bonding, generally leads to higher melting point.
What are the reasons why do we have to determine the chemical properties of materials?
Chemical properties can be used for building chemical classifications. They can also be useful to identify an unknown substance or to separate or purify it from other substances. Materials science will normally consider the chemical properties of a substance to guide its applications.
Why is it helpful to know the physical and chemical properties of a substance?
Why is it helpful to know the physical and chemical properties of a substance? Each substance has a particular set of physical and chemical properties. These properties can be used to identify a substance or to predict how it will behave.
Is freezing point intensive or extensive?
Thus, freezing point is an intensive property and doesn’t change when we vary amount. Other examples of intensive properties are color, boiling point, pressure, molecular weight and density.
Why is it important to identify the different properties of an object?
It’s important for scientists to know the properties of matter because all things are made up of matter. Each type of matter has different physical characteristics and scientists need to know and understand these characteristics to make calculations. … The main phases of matter are solid, liquid, and gas.
How can understanding properties help you separate substances from a mixture?
The substances in a mixture are separated by the differences in their physical properties, such as their particle size. The more different the properties are, the easier it is to separate the substances. Tea leaves do not dissolve in water, so you can use a strainer to FILTER them.