Why are esters considered derivatives of carboxylic acids
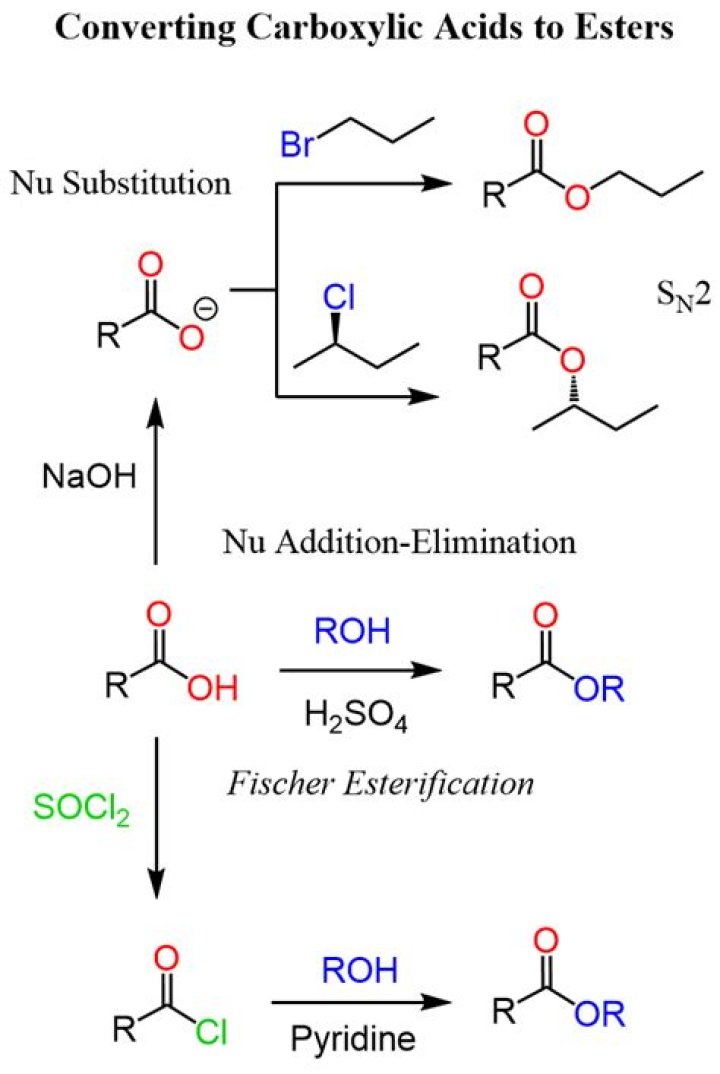
Esters are considered to be functional derivatives of carboxylic acids in which the acidic proton has been replaced by an organic group. The synthesis of esters by the acid catalyzed condensation of a carboxylic acid and an alcohol was considered at the end of the previous chapter.
Why are esters derivatives of carboxylic acids?
Esters and amides are considered to be derivatives of carboxylic acids because the OH in the carboxyl group is replaced with another group.
What is derivatives of carboxylic acid?
The functional groups at the heart of this chapter are called carboxylic acid derivatives: they include carboxylic acids themselves, carboxylates (deprotonated carboxylic acids), amides, esters, thioesters, and acyl phosphates. Cyclic esters and amides are referred to as lactones and lactams, respectively.
Are esters carboxylic acid derivatives?
Esters are one of the most common carboxylic derivatives. … The distinguishing carbonyl substituents in these compounds are: chloro (acyl chlorides), acyloxy (anhydrides), alkoxy (esters) and amino (amides). All of these substituents have bonds originating from atoms of relatively high electronegativity (Cl, O & N).How acid derivatives is produced from carboxylic acid?
Carboxylic acids have a hydroxyl group bonded to an acyl group, and their functional derivatives are prepared by replacement of the hydroxyl group with substituents, such as halo, alkoxyl, amino and acyloxy. The carboxylic acid derivatives can all be hydrolyzed to carboxylic acids.
Why are esters more reactive than carboxylic acids?
Under the reaction condition s the carboxylic acid will deprotonate to give the carboxylate which is a very poor electrophile (after all, it has a negative charge !) so the ester is more reactive than the acid.
Why are carboxylic acids derivatives important?
Carboxylic acids and their derivatives are used in the production of polymers, biopolymers, coatings, adhesives, and pharmaceutical drugs. They also can be used as solvents, food additives, antimicrobials, and flavorings.
Is nitrile a carboxylic acid derivative?
Nitriles of Carboxylic Acids Nitriles contain the cyano group, and although they lack the carbonyl group that the other carboxylic acid derivatives have, they are still classified as carboxylic acid derivatives since they are hydrolyzed to carboxylic acids, and also can be produced by dehydration of primary amides.Which is more reactive between carboxylic acids and carboxylic acid derivatives?
Among the carboxylic acid derivatives, carboxylate groups are the least reactive towards nucleophilic acyl substitution, followed by amides, then carboxylic esters and carboxylic acids, thioesters, and finally acyl phosphates, which are the most reactive among the biologically relevant acyl groups.
Is acid halide a derivative of carboxylic acid?Compounds in which the −OH group of the carboxylic acid is replaced by other functional groups are called carboxylic acid derivatives, the most important of which are acyl halides, acid anhydrides, esters, and amides.
Article first time published onWhich of the following is not a derivative of carboxylic acid?
Which of the following is not a derivative of a carboxylic acid? Explanation: Aldehyde is not a derivative of carboxylic acid. Esters can be derived from carboxylic acids by reacting them with to form an acid chloride.
What is the characteristic reaction of carboxylic acid derivatives?
Nucleophilic acyl substitutions is the characteristic reaction of carboxylic acid derivatives. This reaction occurs with both negatively charged nucleophiles and neutral nucleophiles. Nucleophilic addition to a carbonyl forms a tetrahedral intermediate with two possible leaving groups, Z or Nu.
Are esters reactive?
Ester Hydrolysis Esters are less reactive than acyl halides and acid anhydrides because the alkoxide group is a poor leaving group with its negative charge fully localized on a single oxygen atom.
What is the other product formed when an ester is made by reacting a carboxylic acid and an alcohol?
Esters and water are formed when alcohols react with carboxylic acids. This reaction is called esterification, which is a reversible reaction. … Since esterification is a reversible reaction, esters can undergo hydrolysis to form corresponding alcohol and organic acid.
How do you turn a carboxylic acid into an ester?
Conversion of carboxylic acids to esters using acid and alcohols (Fischer Esterification) Description: When a carboxylic acid is treated with an alcohol and an acid catalyst, an ester is formed (along with water). This reaction is called the Fischer esterification.
What are the primary use of esters?
The main use of esters is for flavourings and perfumes, however they can also be used in the chemicals industry as solvents.
What is Ester in chemistry?
An ester is a chemical compound derived from an acid (organic or inorganic) in which at least one –OH hydroxyl group is replaced by an –O– alkyl (alkoxy) group, as in the substitution reaction of a carboxylic acid and an alcohol.
What are acid derivatives definition?
Carboxylic acid derivative: A carbonyl-containing functional group other than an aldehyde or ketone, or a molecule containing such a functional group. Called a carboxylic acid derivative because these functional groups can, in principle, be produced (derived) from the corresponding carboxylic acid.
Why are esters less reactive than carboxylic acids?
Carboxylic acids and esters have delocalization of electrons. This provides extra stability. That’s why they are less reactive to nucleophiles than aldehyde and ketones.
Why are esters less acidic than ketones?
Esters are more acidic than ketones, because the resonance between the two oxygen atoms gives less opportunity for the delocalization of the electron pair on the alpha carbon in esters contrary to what happens in ketones.
Are esters or thioesters more reactive?
A thioester is more reactive than an ester, for example, because a thiolate (RS-) is a weaker base and better leaving group than an alcoxide (RO-). … Thioesters, for example, are often converted directly into carboxylic esters in biochemical reactions, but not the other way around.
Which one of these carboxylic acid derivatives is the most reactive?
Acyl chlorides are the most reactive carboxylic acid derivatives.
What is the reactivity order of derivatives of carboxylic acid?
Acid Chloride > Amide > Acid Anhydride > Ester.
Why are esters reactive?
Esters are fairly reactive due to the electrophilicity of the C=O. carbon and to its capacity to stabilize alpha enolate anions. This reaction is the reverse of Fischer’s esterification and its mechanism is the same but inverting the sequence (Principle of Microscopic Reversibility).
How do you turn a carboxylic acid into a nitrile?
Nitriles can be converted to carboxylic acid with heating in sulfuric acid. During the reaction an amide intermediate is formed.
Is nitrile more reactive than carboxylic acid?
Structures and reactivity of carboxylic acid derivatives Esters are less reactive than anhydrides; amides and nitriles are less reactive than esters. Hydrolysis of any carboxylic acid derivative gives a carboxylic acid, which can be converted to any other carboxylic acid via the acid chloride.
Why are esters less reactive than acid chloride?
Thus, in comparison to esters, where the oxygen atom need only stabilize one carbonyl group, anhydrides are more reactive than esters. Acid chlorides are very much less stable because chlorine is not effective at stabilizing positive charge by donation of electron density by resonance.
Which of the FF is the carboxylic acid derivative produced when two molecules of carboxylic acid lose water?
The acid anhydride functional group results when two carboxylic acids combine and lose water (anhydride = without water). Symmetrical acid anhydrides are named like carboxylic acids except the ending -acid is replaced with -anhydride. This is true for both the IUPAC and Common nomenclature.
What catalyst is used in making esters?
Esters occur naturally – often as fats and oils – but they can be made in the laboratory by reacting an alcohol with an organic acid . A little sulfuric acid is needed as a catalyst .
Which of the following is not a carboxylic compound?
Picric acid is trinitrophenol and does not have carboxyl group .
What are the first 4 carboxylic acids?
The first four carboxylic acids derived from alkanes are methanoic acid (HCOOH), ethanoic acid (CH3COOH), propanoic acid (C2H5COOH) and butanoic acid (C3H7COOH).



