Which particles are easier to add or remove from an atom
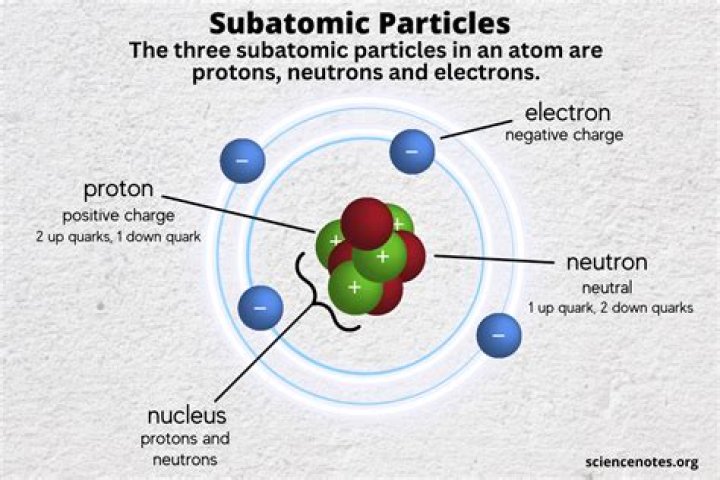
In general, electrons are easier to add or remove from an atom than a proton or neutron.
Which particles can be added or removed from an atom?
Protons carry a positive electrical charge and they alone determine the charge of the nucleus. Adding or removing protons from the nucleus changes the charge of the nucleus and changes that atom’s atomic number. So, adding or removing protons from the nucleus changes what element that atom is!
Is it easier to remove an electron from a small atom?
Each orbital can only contain a limited number of electrons, so for atoms with a large number of protons, the electrons must occupy the orbitals further away from the nucleus. The farther away an electron is from the nucleus, generally then, it will be easier to remove from the atom.
Which electron is the easiest to remove from an atom?
Valence electrons are the farthest from the positive charge (the protons) and thus tend to be easier to remove than core electrons; this means that it takes them less energy to move far away from the atom.What does adding a neutron do?
Atoms always have an equal number of protons and electrons, and the number of protons and neutrons is usually the same as well. Adding a proton to an atom makes a new element, while adding a neutron makes an isotope, or heavier version, of that atom.
Which particles are difficult to remove from an atom which particles are easier to add or remove from an atom?
In general, electrons are easier to add or remove from an atom than a proton or neutron. Chemical reactions largely involve atoms or groups of atoms and the interactions between their electrons.
What happens when an electron is added to an atom?
When electrons are added to an atom, the increased negative charge puts stress on the electrons already there, causing energy to be released. When electrons are removed from an atom, that process requires energy to pull the electron away from the nucleus. Addition of an electron releases energy from the process.
Why is it harder to remove an electron from a full shell?
It becomes harder to remove an electron when an atom has a net positive charge because the attraction that the nuclear charge exerts per electron gets larger. For example, if you have a neutral nitrogen atom, it has 7 electrons.Which electron is the hardest to remove?
The ionization energy of the elements increases as one moves up a given group because the electrons are held in lower-energy orbitals, closer to the nucleus and thus more tightly bound (harder to remove). Based on these two principles, the easiest element to ionize is francium and the hardest to ionize is helium.
Would it be harder or easier to take an electron away from them?The more electrons that are lost, the more positive this ion will be, the harder it is to separate the electrons from the atom. In general, the further away an electron is from the nucleus, the easier it is for it to be expelled.
Article first time published onWould it be harder or easier to remove an electron from an element further down in a group?
Down a group, the number of energy levels (n) increase and the distance is greater between the nucleus and highest-energy electron. The increased distance weakens the nuclear attraction to the outer-most electron, and is easier to remove (requires less energy).
What happens if you remove a neutron?
When you remove or add a neutron to the nucleus of an atom, the resulting substance is a new type of the same element and is called an isotope. Everything you see around you is made up of matter, and all matter is made up of atoms. Can atoms be further broken down?
Is it possible to remove protons from an atom?
Yes We can remove a proton or a neutron from the nucleus of an atom by supplying energy equal to or greater than the seperation energy of the neucleon to be removed.
How does the addition and subtraction of subatomic particles affect the atom?
If you add or subtract a proton from the nucleus, you create a new element. If you add or subtract a neutron from the nucleus, you create a new isotope of the same element you started with. In a neutral atom, the number of positively charged protons in the nucleus is equal to the number of orbiting electrons.
How are electrons removed from an atom?
Ionization is the process in which one or more electrons are removed from an atom or molecule, thereby creating an ion. … In order to remove an electron from an atom, enough energy must be supplied to break the bond between the negatively charged electron and the positively charged nucleus; this is the ionization energy.
When an electron is removed from an atom its energy?
Energy is supplied in order to remove electron from an atom, so energy of atom increases when electron is removed from an atom.
When electrons are removed from an atom then it is called?
Simplified ionization process where an electron is removed from an atom. Ionization is the process by which ions are formed by gain or loss of an electron from an atom or molecule.
What particle would transfer between the atoms the easiest?
The transfer of electrons results in what is known as an ionic bond between atoms, while sharing of electrons creates a covalent bond.
Are valence electrons the most difficult to remove?
Valence electrons are most difficult of all electrons to remove. Core electrons are the easiest of all electrons to remove. … An orbital that penetrates into the region occupied by core electrons is less shielded from nuclear charge than an orbital that does not penetrate and therefore has a lower energy.
How much more energy does it require to remove a proton compared to an electron?
In general, the binding energy of a single proton or neutron in a nucleus is approximately a million times greater than the binding energy of a single electron in an atom.
Why is it easier to remove an electron from B than be?
This is not a linear trendm the ionisation energy of boron being unexpectedly less than that for beryllium, but this is due to the 2s orbital being totally filled in beryllium, whereas boron has one electron in a 2p orbital as well, and the 2s orbitalis shielded much more than the 2p orbital, which gives boron a lower …
Which is easier to remove an electron from B or be?
The general trend of increasing ionization energy across the table is broken at two points. Boron has a smaller value than beryllium, and oxygen has a smaller value than nitrogen. … Thus the 2p electron in boron is easier to remove than a 2s electron in beryllium.
Why is it easier to remove an electron from oxygen than nitrogen?
Since there are two electrons in the first orbital of the O sublevel, there is greater electronic repulsion in the 2p sublevel for O than N. Therefore, it is easier to remove an electron from the O than the N, and the ionization energy of O is lower than N.
Is it easier or harder to remove a second electron from an atom?
For a second level electron as compared to a third level electron, the average force experienced and also the distance for which the electron has to move against that force (until the force becomes negligible) are both larger. Therefore it takes more energy and is harder to remove.
Why is it easier to remove electrons that are farther away from the nucleus?
Electrons closest to the nucleus will try to be as close to the nucleus as possible. The outer/valence electrons that are farther away from the nucleus will be shielded by the inner electrons. … That is why it is easier to remove valence electrons than the inner electrons. It also reduces the nuclear charge of an atom.
Is it easier to remove an electron from H or he?
The first ionization energy for helium is slightly less than twice the ionization energy for hydrogen because each electron in helium feels the attractive force of two protons, instead of one. It takes far less energy, however, to remove an electron from a lithium atom, which has three protons in its nucleus.
Why do nonmetals gain electrons?
Nonmetals want to gain electrons because they have more valence electrons than metals, so it is easier for them to gain electrons than lose the valance electrons to fulfill a stable octet. In addition, nonmetals’ valance electrons are closer to the nucleus, thus allowing more attraction between the two.
Which is lighter electron or proton?
electron, lightest stable subatomic particle known. It carries a negative charge of 1.602176634 × 10−19 coulomb, which is considered the basic unit of electric charge. The rest mass of the electron is 9.1093837015 × 10−31 kg, which is only 1/1,836the mass of a proton.
Which particle if lost from the nucleus will not result in a change in the atomic mass number?
Therefore, when a nucleus loses a neutron the atomic number does not change.
What happens to an atom that loses a neutron or electron?
This happens when an atom gains or loses electrons. … Once an atom has an electrical charge it is called an ion. In an ion the atomic number and atomic mass do not change from the original. If an atom were to gain or lose neutrons it becomes an isotope.
Which particles protons neutrons electrons can be removed or added to an atom?
Yes, we can remove protons and neutrons from an atom. When protons and neutrons combines to form a nucleus, their combined mass is less than that of the sum of their individual masses. This is due to the loss of energy (in achieving a more stable configuration) called binding energy.



