Which law describes the proportional relationship between the pressure and volume of gas
This relationship between pressure and volume is known as Boyle’s law, after its discoverer, and can be stated as follows: At constant temperature, the volume of a fixed amount of a gas is inversely proportional to its pressure.
What is the relationship between pressure and volume of a gas?
The Relationship between Pressure and Volume: Boyle’s Law As the pressure on a gas increases, the volume of the gas decreases because the gas particles are forced closer together. Conversely, as the pressure on a gas decreases, the gas volume increases because the gas particles can now move farther apart.
Is pressure and volume directly proportional?
For a fixed mass of an ideal gas kept at a fixed temperature, pressure and volume are inversely proportional. Or Boyle’s law is a gas law, stating that the pressure and volume of a gas have an inverse relationship. If volume increases, then pressure decreases and vice versa, when the temperature is held constant.
Which law describes the relationship between the pressure and volume of a gas quizlet?
Boyle’s Law describes the relationship between P(pressure) and V (volume) of gases.What gas law states that volume and pressure are inversely proportional while directly proportional to temperature when moles are held constant?
Boyle’s Law – states that the volume of a given amount of gas held at constant temperature varies inversely with the applied pressure when the temperature and mass are constant.
What law describes the relationship between temperature and pressure?
Gay-Lussac’s law is the gas law that describes the pressure-temperature relationship. It states that, at a constant volume, the pressure of a given amount of a particular gas is directly proportional to its Kelvin temperature. … P/T = k where k is a constant, or. P1/T1 = P2/T.
Which statement is a description of Charles's law?
Charles’s law, a statement that the volume occupied by a fixed amount of gas is directly proportional to its absolute temperature, if the pressure remains constant. This empirical relation was first suggested by the French physicist J.
How is the relationship between gas pressure and gas volume different from the relationship between gas volume and gas temperature?
The volume of a given gas sample is directly proportional to its absolute temperature at constant pressure (Charles’s law). The volume of a given amount of gas is inversely proportional to its pressure when temperature is held constant (Boyle’s law).What is the relationship between pressure and volume according to Boyle's Law?
This empirical relation, formulated by the physicist Robert Boyle in 1662, states that the pressure (p) of a given quantity of gas varies inversely with its volume (v) at constant temperature; i.e., in equation form, pv = k, a constant.
What is Charles Law and Boyle's law?Boyle’s law—named for Robert Boyle—states that, at constant temperature, the pressure P of a gas varies inversely with its volume V, or PV = k, where k is a constant. … Charles (1746–1823)—states that, at constant pressure, the volume V of a gas is directly proportional to its absolute (Kelvin) temperature T, or V/T = k.
Article first time published onWhat is Boyle's and Charles Law?
Boyle showed that the volume of a sample of a gas is inversely proportional to its pressure (Boyle’s law), Charles and Gay-Lussac demonstrated that the volume of a gas is directly proportional to its temperature (in kelvins) at constant pressure (Charles’s law), and Avogadro postulated that the volume of a gas is …
Are pressure and volume inversely proportional?
When the volume decreases, the pressure increases. This shows that the pressure of a gas is inversely proportional to its volume. This is shown by the following equation – which is often called Boyle’s law. … It means that for a gas at a constant temperature, pressure × volume is also constant.
Are pressure and temperature inversely proportional?
Pressure is inversely proportional to volume when the temperature is held constant for a given amount of gas. It is also known as Boyle’s law. Pressure is directly proportional to the temperature when the volume is held constant for a given amount of gas. It is also known as Gay-Lussac law.
Which of the following best describes the inverse relationship of volume and pressure of gases at constant temperature?
Boyle’s Law describes the inverse relationship between the pressure and volume of a fixed amount of gas at a constant temperature.
What is the relationship described by Charles's law assuming all the other variables are held constant?
The volume of a given gas sample is directly proportional to its absolute temperature at constant pressure (Charles’s law). The volume of a given amount of gas is inversely proportional to its pressure when temperature is held constant (Boyle’s law).
Which of the following relationship represents Charle's law?
Notes: When the pressure is held constant, the volume of a quantity of the gas is related to the temperature as V/T = constant. This relationship is known as Charles’ law.
What does Avogadro's law state?
Avogadro’s law, a statement that under the same conditions of temperature and pressure, equal volumes of different gases contain an equal number of molecules. … The law is approximately valid for real gases at sufficiently low pressures and high temperatures.
Which empirical gas law describes the relationship between the volume and pressure of a gas when the number of moles and temperature are constant?
Avogadro’s law states that “equal volumes of all gases, at the same temperature and pressure, have the same number of molecules.” For a given mass of an ideal gas, the volume and amount (moles) of the gas are directly proportional if the temperature and pressure are constant.
What is the relationship between volume and pressure?
Volume and Pressure: Boyle’s Law Decreasing the volume of a contained gas will increase its pressure, and increasing its volume will decrease its pressure. In fact, if the volume increases by a certain factor, the pressure decreases by the same factor, and vice versa.
Which gas law relates volume and pressure express the law mathematically?
The relationship for Boyle’s Law can be expressed as follows: P1V1 = P2V2, where P1 and V1 are the initial pressure and volume values, and P2 and V2 are the values of the pressure and volume of the gas after change.
How does Boyle's law describe the relationship between gas pressure and volume quizlet?
How does Boyle’s law describe the relationship between gas pressure and volume? 3. At a constant temperature (when the temp doesn’t change) when gas pressure is increased gas volume decreases. This is an inverse relationship (because as one (pressure) gets bigger the other (volume) gets smaller or vice versa).
Which gas law is inversely proportional?
Boyle’s law He observed that volume of a given mass of a gas is inversely proportional to its pressure at a constant temperature. Boyle’s law, published in 1662, states that, at constant temperature, the product of the pressure and volume of a given mass of an ideal gas in a closed system is always constant.
Which laws can be combined to form the ideal gas law?
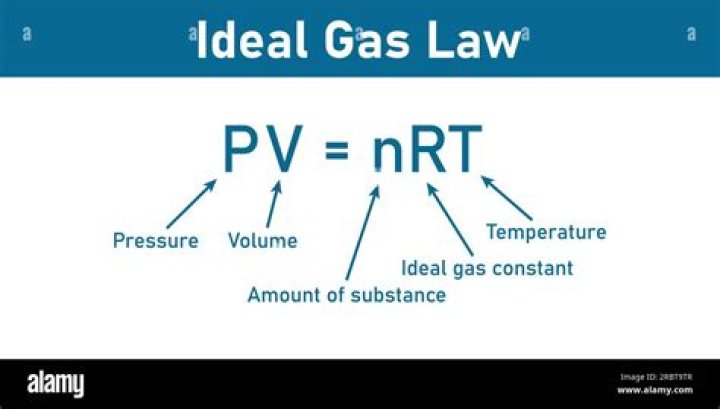
The combined gas law combines the three gas laws: Boyle’s Law, Charles’ Law, and Gay-Lussac’s Law. It states that the ratio of the product of pressure and volume and the absolute temperature of a gas is equal to a constant. When Avogadro’s law is added to the combined gas law, the ideal gas law results.
Which two variables are compared in Charles's law?
Since pressure is kept constant, the only variable that is manipulated is temperature. This means that we can use Charles’s law in order to compare volume and temperature. Since volume and temperature are on opposite sides of the ideal gas law, they are directly proportional to one another.
Why is gas pressure and volume inversely proportional?
Boyle’s Law is a relationship between pressure and volume. In this relationship, pressure and volume have an inverse relationship when temperature is held constant. … If there is an increase in the volume the molecules have more space to move, collisions happen less often and the pressure is decreased.
Is the relationship between the pressure and volume of a confined gas direct or inverse?
The relationship between the pressure and volume of a confined gas is inversely proportional. The data collected and the graphs plotted prove this relationship. The shapes of the curves in the graphs and the fact that when the volume decreases, the pressure increases (vice-versa), also prove the inverse relationship.
What does inversely mean in Boyle's law?
Boyle’s law states that the volume of a given mass of gas varies inversely with the pressure when the temperature is kept constant. An inverse relationship is described in this way. As one variable increases in value, the other variable decreases.
Is Charles Law direct or inverse?
Charles Law is a direct relationship between temperature and volume. When the temperature of the molecules increases the molecules move faster creating more pressure on the container of the gas increasing the volume, if the pressure remains constant and the number of the molecules remains constant.



