What makes a particle positively charged
The matter is positively charged if it contains more protons than electrons, and it is negatively charged if it contains more electrons than protons. In both instances, charged particles will experience a force when in the presence of other charged matter.
What is a positively charged particle?
Protons are a type of subatomic particle with a positive charge. Protons are bound together in an atom’s nucleus as a result of the strong nuclear force. Neutrons are a type of subatomic particle with no charge (they’re neutral).
What makes a particle positive or negative?
The most common charged particles are the electron and proton. Atoms with an excess of electrons are called negative ions. Those with missing electrons are called positive ions.
What gives a particle charge?
Electric charge is carried by subatomic particles. In ordinary matter, negative charge is carried by electrons, and positive charge is carried by the protons in the nuclei of atoms. … The proton has a charge of +e, and the electron has a charge of −e.What is alpha particle charge?
alpha particle, positively charged particle, identical to the nucleus of the helium-4 atom, spontaneously emitted by some radioactive substances, consisting of two protons and two neutrons bound together, thus having a mass of four units and a positive charge of two.
Which of the following particle has positive charge?
proton, stable subatomic particle that has a positive charge equal in magnitude to a unit of electron charge and a rest mass of 1.67262 × 10−27 kg, which is 1,836 times the mass of an electron.
How do you know if an atom is positive or negative?
If the atom has more electrons than protons, it is a negative ion, or ANION. If it has more protons than electrons,it is a positive ion.
What particle has negative charge?
electron, lightest stable subatomic particle known. It carries a negative charge of 1.602176634 × 10−19 coulomb, which is considered the basic unit of electric charge. The rest mass of the electron is 9.1093837015 × 10−31 kg, which is only 1/1,836the mass of a proton.Can electrons be positive?
Electrons. … Proton—positive; electron—negative; neutron—no charge. The charge on the proton and electron are exactly the same size but opposite. The same number of protons and electrons exactly cancel one another in a neutral atom.
Why are alpha particles positively charged?An alpha particles is positively charged because it is essentially the nucleus of a Helium-4 atom. A Helium-4 nucleus is composed of two protons, which are positively charged particles, and two neutrons, which have no electric charge.
Article first time published onDo alpha particles have a positive charge?
A positively charged particle ejected spontaneously from the nuclei of some radioactive elements. It is identical to a helium nucleus that has a mass number of 4 and an electrostatic charge of +2.
Are beta particles positively charged?
A negatively charged beta particle is identical to an electron, while a positively charged beta particle is called a positron. … Large amounts of beta radiation may cause skin burns, and beta emitters are harmful if they enter the body. Beta particles may be stopped by thin sheets of metal or plastic.
How can u tell if an atom has a positive charge?
The matter is positively charged if it contains more protons than electrons, and it is negatively charged if it contains more electrons than protons.
Are atoms positive neutral or negative?
Atoms are neutral; they contain the same number of protons as electrons. By definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion.
What is a rule for making a positive ion which has positive charge?
Elements on the left-most part of the periodic table of elements have one extra electron to the octet rule. This is why they want to give an electron away to something else, like a flourine atom. When the element gives off an electron, it gets a positive charge, making it a positive ion.
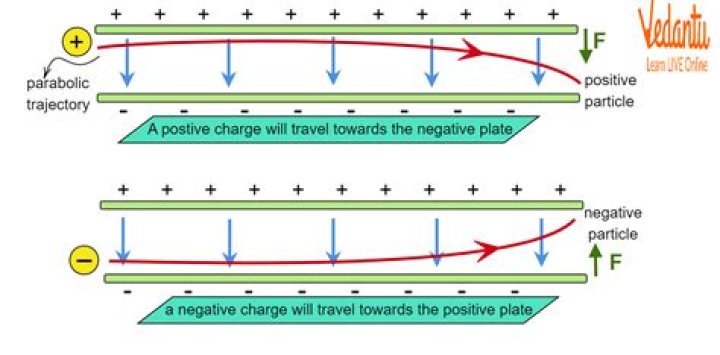
Why are electron particles negatively charged?
So, the electrons were deflected towards the positive plate because there is a negative charge on the electrons. This negative charge on the electron makes it a negative particle.
Is the nucleus positively charged?
Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. … The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral.
Is neutron a positive charge?
Among atomic particles, the neutron seems the most aptly named: Unlike the positively charged proton or the negatively charged electron, neutrons have a charge of zero.
Are humans negatively or positively charged?
The human body is neutrally charged. You feel static charge because the hairs on your skin gets positively or negatively charged when rubbed.
What gives an electron its charge?
So now we can answer your question in the language of the quantum field: the electron gets its charge by the field allowing to create one positive charge state and one negative charge state at the same time, leaving its total charge zero.
Is a gamma particle positively charged?
Alpha particles are positively charged, beta particles are negatively charged, and gamma radiation is electrically neutral .
Why does the alpha particle have a 2+ charge?
An alpha particle is identical to a helium atom that has been stripped of its two electrons; thus, an alpha particle contains two protons and two neutrons. Because an alpha particle has no electrons to balance the positive charge of the two protons, it has a charge of +2 and can be represented as He2+.
Why are alpha particles doubly charged?
alpha particles are helium nuclei i.e. it has lost two electron thus it has 2 protons and 2 neutrons. Because a proton has a charge of +1, the nuclei as a whole has charge +2 so an alpha particle is doubly positively charged.
Do alpha particles have a negative charge?
Alpha particles (α) are positively charged and made up of two protons and two neutrons from the atom’s nucleus.
Is Beta positive or negative charge?
Beta particles are negatively charged electrons emitted by the nucleus on decay (splitting of a neutron).
Do beta particles have a charge of 2+?
Beta particles have a charge of minus 1, and a mass of about 1/2000th of a proton. This means that beta particles are the same as an electron. … This means that they have no mass and no charge.
What is the difference between beta and electron?
Main Difference – Beta Particle vs Electron Electrons are subatomic particles that can be found in the electron cloud that surrounds the atomic nucleus. The main difference between beta particle and electron is that beta particle can have either +1 charge or -1 charge whereas electron has a -1 charge.
What is a beta particle made up of?
Beta decay A beta particle forms when a neutron changes into a proton and a high-energy electron . The proton stays in the nucleus but the electron leaves the atom as a beta particle.
How does an atom become a positive ion How does an atom become a negative ion?
Ions form when atoms gain or lose electrons. Since electrons are negatively charged, an atom that loses one or more electrons will become positively charged; an atom that gains one or more electrons becomes negatively charged.
What makes the nucleus of the atom positively charged?
Explanation: A nucleus of an atom has protons and neutrons. We know that a proton has a charge of +1 , while a neutron has no charge, or 0 . Therefore, the nucleus of an atom will always have a positive charge.



