What is the temperature at which the vapor pressure is equal to the external pressure on a liquid called
The boiling point of a liquid is the temperature at which its vapor pressure is equal to the pressure of the gas above it. The normal boiling point of a liquid is the temperature at which its vapor pressure is equal to one atmosphere (760 torr).
When the external pressure is equal to the vapor pressure of a liquid?
When the vapor pressure equals the external pressure, bubbles of vapor form within the liquid, and it boils. The temperature at which a substance boils at a pressure of 1 atm is its normal boiling point.
Why does boiling occur when vapor pressure equals atmospheric pressure?
When any substance in the liquid phase, at atmospheric pressure, it means that this pressure is sufficient to confine the molecules together to form liquid. The molecules should possess equal energy to overcome this pressure for the liquid to boil.
Is vapor pressure the same as external pressure?
It is important to note that when a liquid is boiling, its vapor pressure is equal to the external pressure. For example, as water boils at sea level, its vapor pressure is 1 atmosphere because the external pressure is also 1 atmosphere.Is vapor pressure proportional to temperature?
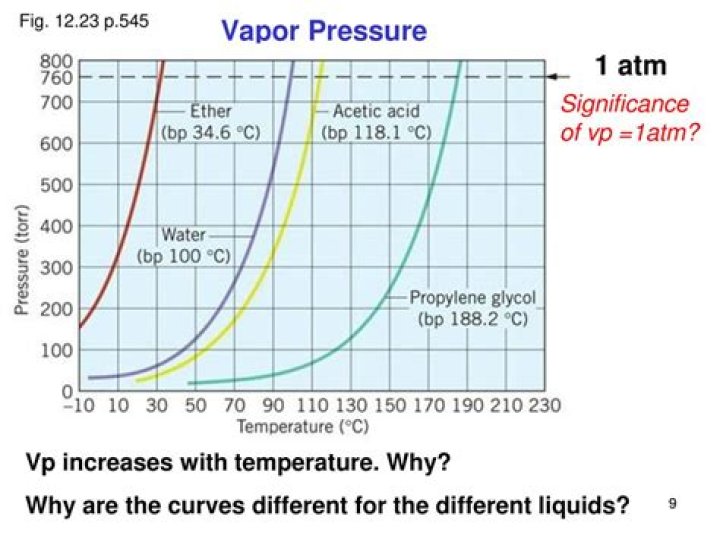
The vapor pressure of a liquid varies with its temperature, as the following graph shows for water. … As the temperature of a liquid or solid increases its vapor pressure also increases. Conversely, vapor pressure decreases as the temperature decreases.
How do you find vapor pressure from temperature?
In chemistry, vapor pressure is the pressure that is exerted on the walls of a sealed container when a substance in it evaporates (converts to a gas). To find the vapor pressure at a given temperature, use the Clausius-Clapeyron equation: ln(P1/P2) = (ΔHvap/R)((1/T2) – (1/T1)).
Why does vapor pressure increase with temperature?
As the temperature of a liquid increases, the kinetic energy of its molecules also increases and as the kinetic energy of the molecules increases, the number of molecules transitioning into a vapor also increases, thereby increasing the vapor pressure.
What is a external pressure?
External pressure occurs when you feel coerced by external forces, like fear of punishment or obsession with attaining rewards, to do your job a certain way. These are different from internal pressures in that you do not typically have control over them.Does external pressure affect temperature?
There is actually no relationship between the two. The air pressure is a measure of the pressure of the air. The vapour pressure of a liquid, say, water, is a function of its temperature and does not depend on the air pressure.
What happens when vapor pressure is equal to atmospheric pressure?When the atmospheric pressure is equal to the vapor pressure of the liquid, boiling will begin. When a liquid boils, what is inside the bubbles? The bubbles in a boiling liquid are made up of molecules of the liquid which have gained enough energy to change to the gaseous phase.
Article first time published onWhen vapor pressure is the same as atmospheric pressure?
Video: Boiling, Atmospheric Pressure, and Vapor Pressure Boiling occurs when the atmospheric pressure and the vapor pressure are equal. The bubbles in a boiling liqued are NOT air. They are the liquid that has changed states to a gas. In boiling water the bubbles are made up of water molecules in a gaseous state.
When Vapour pressure is lower than atmospheric pressure boiling occurs?
If the vapor pressure is lower than the atmospheric pressure, it means that the thermal energy of molecules is not strong enough for them in the liquid to break free to enter gas phase. Liquid molecules can not form bubbles of vapor within the liquid. Therefore boiling would never occur.
Are temperature and vapor pressure linearly proportional?
The vapor pressure of any substance increases non-linearly with temperature according to the Clausius–Clapeyron relation. The atmospheric pressure boiling point of a liquid (also known as the normal boiling point) is the temperature at which the vapor pressure equals the ambient atmospheric pressure.
Which has maximum vapour pressure at a given temperature?
Ether has the highest vapour pressure (i.e., lowest boilint point) at any temperature. The reason for its high vapour pressure is that the attraction is less between ether molecules than between water and alcohol molecules.
Which has lowest vapor pressure?
The substance with the highest boiling point will have the lowest vapor pressure. Vapor pressure is a liquid property related to evaporation. In the liquid (or any substance) the molecules have a distribution of kinetic energies related to the temperature of the system.
Does pressure increase with temperature?
Faster moving particles will collide with the container walls more frequently and with greater force. This causes the force on the walls of the container to increase and so the pressure increases. If the temperature of the gas is measured on the Kelvin scale , the pressure is proportional to the temperature.
Which of the following has the highest vapor pressure?
Among the choices, liquid ether molecules more readily escape as vapor so that the boiling point is reached sooner. At 25°C, ether has the highest vapor pressure.
What is the temperature when water and vapor are in equilibrium with the atmospheric pressure?
If the liquid is in an open container and exposed to normal atmospheric pressure, the liquid boils when its saturated vapor pressure becomes equal to 1 atmosphere (or 101325 Pa or 101.325 kPa or 760 mmHg). This happens with water when the temperature reaches 100°C.
What is the vapor pressure of A at 35 C?
T °CP (torr)3542.23644.63747.13849.7
How do you calculate total vapor pressure?
The total vapour pressure (p) is a linear function of the mole fraction xB (or xA as xA =1-xB ) because pA° and pB° are constant at a particular temperature. The dotted lines give the partial pressure of the two components versus composition and the solid line gives the vapour pressure versus composition.
Why does vapor pressure only depend on temperature?
Vapor pressure is dependent upon temperature. When the liquid in a closed container is heated, more molecules escape the liquid phase and evaporate. The greater number of vapor molecules strike the container walls more frequently, resulting in an increase in pressure.
What is meant by the term vapor pressure how it is affected by external pressure?
Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with temperature. The temperature at which the vapour pressure at the surface of a liquid becomes equal to the pressure exerted by the surroundings is called the boiling point of the liquid.
What is external hydrostatic pressure?
[ hī′drə-stăt′ĭk ] The pressure exerted by a fluid at equilibrium at a given point within the fluid, due to the force of gravity. Hydrostatic pressure increases in proportion to depth measured from the surface because of the increasing weight of fluid exerting downward force from above.
How does the internal pressure compared to the external pressure science?
The internal pressure is either equal, or not equal, to the external pressure. If the work done by the system is equal in magnitude to the work done by the surroundings (which you confirmed was true), the magnitude of the force applied by the system must equal the magnitude of the force applied by the surroundings.
Is peer pressure an external pressure?
What is peer pressure? Peer pressure is internal or external pressure felt to behave in certain ways, both good and bad. … Changing hormones, developing brains and emerging identities makes the start of adolescence a particularly vulnerable time, where peer pressure is most influential.
When the air temperature increases the saturation vapor pressure will ____?
when the air temperature increases, the saturation vapor pressure decreases.
What is vapor pressure of water at 25 degrees Celsius?
The vapor pressure of water at room temperature (25° C) is 0.0313 atm, or 23.8 mm of mercury (760 mm Hg = 1 atm).
What is the temperature when the water boils answer?
At sea level, water boils at 100° C (212° F). At higher altitudes the temperature of the boiling point is lower.
What is the temperature when the water boils?
A liquid at high pressure has a higher boiling point than when that liquid is at atmospheric pressure. For example, water boils at 100 °C (212 °F) at sea level, but at 93.4 °C (200.1 °F) at 1,905 metres (6,250 ft) altitude. For a given pressure, different liquids will boil at different temperatures.
Is vapor pressure the same as boiling point?
The boiling point of a liquid is the temperature at which its vapor pressure is equal to the pressure of the gas above it. The normal boiling point of a liquid is the temperature at which its vapor pressure is equal to one atmosphere (760 torr).
On what factors does Vapour pressure depends?
The liquid’s vapour pressure depends on the nature of the fluid and temperature, with an increase of intermolecular force of attraction Vapour pressure of liquid decreases, and with a rising temperature vapour pressure of liquid increases.