What is the conclusion of mixing baking soda and vinegar
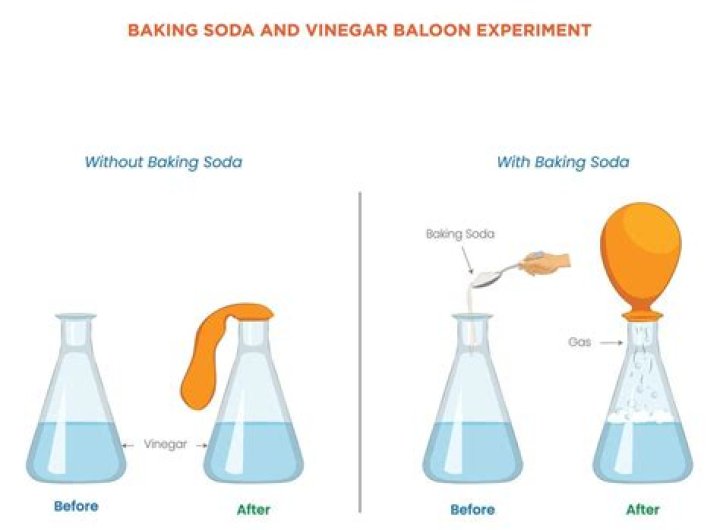
The experiment and result of it supported our hypothesis that the bubbles would float on top of the mixture of the baking soda and vinegar. It did this because when we combined the baking soda and vinegar it had a chemical reaction that produces carbon dioxide gas.
What can be concluded about mixing vinegar and baking soda?
When baking soda is mixed with vinegar, something new is formed. The mixture quickly foams up with carbon dioxide gas. If enough vinegar is used, all of the baking soda can be made to react and disappear into the vinegar solution. … Sodium bicarbonate and acetic acid reacts to carbon dioxide, water and sodium acetate.
What type of reaction is the reaction of baking soda and vinegar?
When vinegar and baking soda are first mixed together, hydrogen ions in the vinegar react with the sodium and bicarbonate ions in the baking soda. The result of this initial reaction is two new chemicals: carbonic acid and sodium acetate. The second reaction is a decomposition reaction.
What is the reason behind baking soda and vinegar mixture reaction?
Reaction Explained Simply The water in the vinegar acts as a host where the base and acid react. During the reaction, when the baking soda is mixed with the vinegar, the baking soda (Base) takes a proton from the vinegar (Acid). The reaction causes the baking soda to transform into water and carbon dioxide.How can you conclude that the reaction is complete?
A reaction is “completed” when it has reached equilibrium — that is, when concentrations of the reactants and products are no longer changing. If the equilibrium constant is quite large, then the answer reduces to a simpler form: the reaction is completed when the concentration of a reactant falls to zero.
What is vinegar and how does it influence the chemical reactions taking place?
Vinegar is a solution of acetic acid in water. Acetic acid reacts with sodium bicarbonate to produce carbon dioxide, just as the citric acid in an effervescent tablet does.
What is the balanced equation for baking soda and vinegar?
Balancing Strategies: In this reaction we have NaHCO3 (baking soda) reacting with an aqueous solution of HC2H3O2 (vinegar) to form NaC2H3O2 + CO2 + H2O.
How do you tell if a reaction will go to completion?
A chemical reaction goes to completion when one of the reactants is used up completely. A chemical reaction is composed of reactants which interact to form products. The presence of each reactant is crucial if a product is to be formed.What happens when baking soda is added to vinegar write a word equation for this reaction name the type of change which takes place?
The equation for the reaction is: CH3COOH + NaHCO3 = CH3COONa + CO2 + H2O. Baking soda and vinegar react with each other because they exchange atoms.
How do you know if a reaction is complete or incomplete?Complete combustion takes place in the presence of a sufficient amount of oxygen while an incomplete combustion reaction takes place when there is an insufficient amount of oxygen supply.
Article first time published onWhat observations might indicate that a chemical reaction is complete?
The five conditions of chemical change: color change, formation of a precipitate, formation of a gas, odor change, temperature change.
How do you use vinegar and baking soda?
Here are some recipes to try. Freshen your sink by mixing one part of baking soda with two parts of vinegar. This mixture unlocks an effervescent fizz of carbon dioxide that cleans and freshen drains. Remove hard water stains by placing a vinegar-soaked towel over the affected area.
Will combining water and baking soda result in a chemical reaction will combining vinegar and baking soda result in a chemical reaction?
Baking soda has the chemical name sodium bicarbonate. Vinegar is a combination of water and 5 percent acetic acid. Since both materials contain chemicals, when the two combine there is a chemical reaction. When vinegar and baking soda are mixed, a new chemical called carbonic acid is made.
What factors affect the completion of a reaction?
- surface area of a solid reactant.
- concentration or pressure of a reactant.
- temperature.
- nature of the reactants.
- presence/absence of a catalyst.
What is the equilibrium of a reaction?
chemical equilibrium, condition in the course of a reversible chemical reaction in which no net change in the amounts of reactants and products occurs. … At equilibrium, the two opposing reactions go on at equal rates, or velocities, and hence there is no net change in the amounts of substances involved.
What is meant by the phrase go to completion?
Reactions that go to completion The phrase go to completion means that the forward direction—such as reaction (1) above—continues until all reactants are used up. The product is prevented from breaking down—as in reaction (2) above—to form the original reactants.
How do you write incomplete combustion?
- Incomplete combustion occurs when the supply of air or oxygen is poor. Water is still produced, but carbon monoxide and carbon are produced instead of carbon dioxide.
- In general for incomplete combustion:
- hydrocarbon + oxygen → carbon monoxide + carbon + water.
- propane + oxygen → carbon + water.
What is the name of the product at the end of both reactions?
The substances that go into a chemical reaction are called the reactants, and the substances produced at the end of the reaction are known as the products.
How does combustion happen?
Combustion is a chemical process in which a substance reacts rapidly with oxygen and gives off heat. … Most of the exhaust comes from chemical combinations of the fuel and oxygen. When a hydrogen-carbon-based fuel (like gasoline) burns, the exhaust includes water (hydrogen + oxygen) and carbon dioxide (carbon + oxygen).
How do you describe what happens in a chemical reaction?
In a chemical reaction, the atoms and molecules produced by the reaction are called products. … No new atoms are created, and no atoms are destroyed. In a chemical reaction, reactants contact each other, bonds between atoms in the reactants are broken, and atoms rearrange and form new bonds to make the products.
Which statement proves that a chemical reaction has occurred?
A color change, formation of a precipitate or a gas, or temperature changes are the evidences of a chemical reaction.
What is important about the properties of a substance that has undergone a chemical change?
The substances produced during chemical changes however cannot easily change back into the original substances. The most important thing for your to remember is that in a physical change the composition of a substance does not change and in a chemical change the composition of a substance does change.
Is baking soda and vinegar a homogeneous mixture?
Combining ingredients to bake cookies or a cake forms what is called a “mixture” in cooking. But, a chemical reaction occurs between the ingredients. The final result (cookies or a cake) is a heterogeneous mixture. Combining baking soda and vinegar causes a chemical reaction.
How much baking soda do I mix with vinegar?
Always keep the ratio one-part baking soda to two parts vinegar. The mixture will fizz and bubble, like your favorite middle school volcano science project.
Will baking powder and vinegar react?
Explain that the bubbles the students observed were produced by carbon dioxide gas generated from the reaction with baking soda, a chemical in both of the powders. The other two ingredients in baking powder do not react with vinegar.
What is the importance of understanding the different factors affecting rate of chemical reactions?
How do we monitor rates of reaction? Firstly it’s important to understand what a rate of reaction is. When a reaction occurs, molecules are colliding together with enough energy for reactants to be broken down or changed into a new species known as a product (often there is more than one products).
What is the role of a catalyst?
A catalyst is a substance that can be added to a reaction to increase the reaction rate without getting consumed in the process. Catalysts typically speed up a reaction by reducing the activation energy or changing the reaction mechanism.
Why does rate of reaction increases with rise in temperature?
An increase in temperature causes a rise in the energy levels of the molecules involved in the reaction, so the rate of the reaction increases.