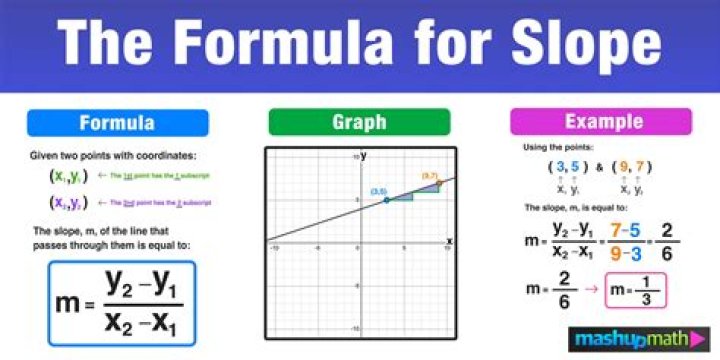
What is an element compound and mixture
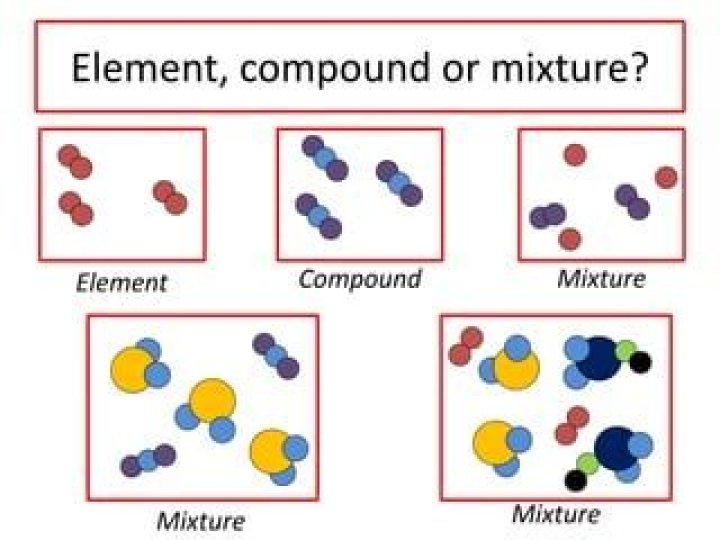
An element contains just one type of atom. A compound contains two or more different atoms joined together. A mixture contains two or more different substances that are only physically joined together, not chemically. A mixture can contain both elements and compounds.
What is difference between element compound and mixture?
Element: Pure substance consisting of one type of atom. Compound: Pure substance consisting of two or more different atoms. Mixture: Two or more different substances not chemically combined.
What is element and compound class 9?
Elements are substances that cannot be separated into simpler substances. A compound is a substance formed when two or more elements are chemically joined. 2. 1 Comments.
What are elements compounds and mixtures examples?
CompoundSymbolElements in the compound (and state at room temp)WaterH2OHydrogen (gas) oxygen (gas)Carbon dioxideCO2Carbon (solid) oxygen (gas)Magnesium oxideMgOMagnesium (solid) oxygen (gas)Iron sulphideFeSIron (solid) Sulphur (solid)What is mixture in chemistry?
A mixture is made when two or more substances are combined, but they are not combined chemically. General properties of a mixture: ● The components of a mixture can be easily separated. ● The components each keep their original properties.
What element is a mixture?
Note that a mixture: consists of two or more different elements and/or compounds physically intermingled, can be separated into its components by physical means, and. often retains many of the properties of its components.
What are elements compounds and mixtures for kids?
- A sample of a pure element contains atoms that are chemically the same, but different from those of all other elements. …
- Compounds are formed by a process called a chemical reaction. …
- Substances such as milk, paint, ink, air, and muddy water are mixtures, as are rocks and metal alloys.
What is element short form?
Short form of elements are symbols. When elements are wittern in form of chemical symbols. For example- Carbon- C. Oxygen-O.What are some examples of a mixture?
- Sand and water.
- Salt and water.
- Sugar and salt.
- Ethanol in water.
- Air.
- Soda.
- Salt and pepper.
- Solutions, colloids, suspensions.
Hint: Element is a pure form of substance which consists of atoms only. Compounds consist of elements in the fixed proportion. Molecules are made up of two or more than two atoms. These atoms may be the same or different.
Article first time published onWhat is element and compound class 6?
Element is the basic unit of matter, which cannot be broken down into more simpler substances by any way but can be combined to form new substances.It is a pure substance and classified into metals, non-metals, metalloids and noble gases. Example – Sulphur, Gold , Iron, Hydrogen, Carbon, Oxygen.
What is mixture explain its types?
Answer: A mixture is a combination of two or more substances (elements or compounds) which is not done chemically. Mixtures are two types Homogeneous Mixtures and Heterogeneous mixtures. Homogeneous Mixtures: A Homogeneous mixture has a uniform composition of its components throughout its mass.
What are the 10 example of mixture?
Examples include ice in water, salt and oil, noodles in broth, and sand and water. Many common liquids are homogeneous mixtures. Examples include dishwashing liquid, shampoo, vinegar, wine, and vodka. Similarly, many common liquids are heterogeneous mixtures.
How do you identify elements and compounds?
Elements are substances that cannot be broken down into simpler substances with different properties. Elements have been organized by their properties to form the periodic table. Two or more atoms can combine to form a molecule. Molecules consisting of more than one element are called compounds.
What is a compound in Science Kid definition?
A compound is two or more different elements chemically bonded together.
What is an element Grade 7?
An element is a pure substance that cannot be broken down by chemical methods into simpler parts. For example. A piece of iron cannot be broken down into anything other than iron.
What is element with example?
This means that all atoms that make up the element would have the same number of protons. For example, carbon is an element comprised of atoms having the same number of protons, i.e. 6. Common examples of elements are iron, copper, silver, gold, hydrogen, carbon, nitrogen, and oxygen.
What is mixture answer?
A mixture is a substance made by combining two or more different materials in such a way that no chemical reaction occurs. A mixture can usually be separated back into its original components. Some examples of mixtures are a tossed salad, salt water and a mixed bag of M&M’s candy.
Is oil a mixture?
Chemical and physical properties Crude oil is a mixture of comparatively volatile liquid hydrocarbons (compounds composed mainly of hydrogen and carbon), though it also contains some nitrogen, sulfur, and oxygen.
What element is Z?
Zirconium – Element information, properties and uses | Periodic Table.
WHAT IS A in chemistry?
A is used to signify the atomic mass number (also known as atomic mass or atomic weight) of an atom. A = # protons + # neutrons.
How many elements are there in science?
The elements of the periodic table sorted by atomic number. click on any elements name for further chemical properties, environmental data or health effects. This list contains the 118 elements of chemistry.
What is an element Class 8?
A substance which cannot be broken down into two or more simpler substances by chemical reactions is called an element. For Example: Iron,hydrogen,helium,oxygen,phosphorus,sulphur,chlorine,bromine,gold,silver,mercury, aluminium etc.
What is an element in science?
chemical element, also called element, any substance that cannot be decomposed into simpler substances by ordinary chemical processes. Elements are the fundamental materials of which all matter is composed.
What is an atom Class 9?
The smallest tiny particles of matter which can’t be divided further is called atom, i.e., an atom is the smallest building block of matter. For example: Sodium (Na), Hydrogen (H), Oxygen (O), etc.
What is an element for Class 4?
An element is a pure substance that cannot be broken down by any physical or chemical means into simpler substances.
What is mixture Class 4?
A mixture is formed when two or more substances are mixed together. A solution is a type of mixture in which one substance completely dissolves in another. An example of a solution is sugar in water. Sand mixed in water forms a mixture but not a solution because both substances retain their physical properties.
What are the elements present in water?
Water molecules are made of hydrogen and oxygen atoms, in the definite proportion of two hydrogens for one oxygen. Hence, a molecule is the smallest subunit of a compound while an atom is the smallest subunit of an element.
Is water a pure substance?
Water, H2O, is a pure substance, a compound made of hydrogen and oxygen. … In distilled water all of the dissolved substances mixed in water have been removed by evaporation. As water evaporates, it distills, or leaves the salt behind. The pure evaporated water is collected and condensed to form distilled water.
Is soap a mixture?
Soap is not a compound, it is a mixture of sodium and potassium salts of long chain fatty acids containing 12 to 18 carbon atoms.
Is toothpaste a mixture?
Toothpaste is a mixture of powdered solids and various liquids, so it’s neither a liquid nor a solid. Chemists would argue that toothpaste is a colloid (like milk or ink): a mixture where tiny particles of one substance are dispersed evenly into another without separating out.