What is a metallic bond example
Answer Not Found
What are examples of metallic substances?
- AgNO3 – Silver nitrate is a metallic compound. Silver (Ag) is the metal, bonded to the nitrate group.
- CaCl2 – Calcium chloride is a metallic compound.
- H2O (water) is not considered a metallic compound. Even though hydrogen sometimes acts like a metal, it is more often considered a nonmetal.
Is NaCl a metallic bond?
Ionic Bonds: Examples include LiF, NaCl, BeO, CaF2 etc. Covalent Bonds: Examples include hydrogen gas, nitrogen gas, water molecules, diamond, silica etc. Metallic Bonds: Examples include iron, gold, nickel, copper, silver, lead etc.
What is a metallic bond simple definition?
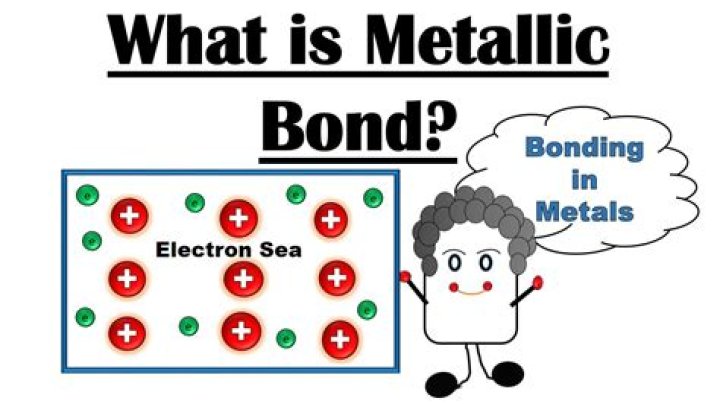
metallic bond, force that holds atoms together in a metallic substance. … The atoms that the electrons leave behind become positive ions, and the interaction between such ions and valence electrons gives rise to the cohesive or binding force that holds the metallic crystal together.What has a metallic bond?
Metallic bonds occur among metal atoms. Whereas ionic bonds join metals to non-metals, metallic bonding joins a bulk of metal atoms. … Metals tend to have high melting points and boiling points suggesting strong bonds between the atoms.
Is copper a metallic bond?
Aluminum foil and copper wire are examples of metallic bonding in action . Aluminum foilA sheet of aluminum foil is made up of metallic bonds. Metallic bonds are mediated by strong attractive forces. This property contributes to the low volatility, high melting and boiling points, and high density of most metals.
What are 5 examples of metallic bonds?
- Sodium (Na) Sodium has a lone electron in its outermost orbital, i.e., the 3s orbital. …
- Magnesium (Mg) Magnesium has two electrons in its outermost shell, the 3s shell. …
- Aluminum (Al) Aluminum has three valence electrons in the 3s orbital.
How do you describe metallic bonding?
Metallic bonding is often described as an array of positive ions in a sea of electrons. The metal is held together by the strong forces of attraction between the delocalised electrons and the positive ions. Beware if you are going to use the term “an array of positive ions in a sea of electrons”!Is steel a metallic bond?
In a metal, each metal atom is conceived to donate a few of its valence electrons to the lattice, such that metal nuclei acquire a positive charge. … So, steel is a metal, and as such displays metallic bonding.
How do you identify metallic bond?A metallic bond is the sharing of many detached electrons between many positive ions, where the electrons act as a “glue” giving the substance a definite structure. It is unlike covalent or ionic bonding. Metals have low ionization energy. Therefore, the valence electrons can be delocalized throughout the metals.
Article first time published onIs aluminum a metallic bond?
Aluminum is non-molecular, and each aluminum nucleus contributes a few of its valence electrons to the metallic lattice. The result is metallic bonding; which gives rise to the usual metallic properties, e.g. malleability, ductility, conductivity towards heat and electricity.
Is iron a metallic bond?
Iron has a type of bonding called metallic bonding. TRUE. Iron is a metal and all metals have a type of bonding called metallic bonding which is different from covalent or ionic bonding. In metallic bonding the outer shells of adjacent atoms overlap, and the outer shell electrons are free to move through the lattice.
Is NaCl ionic or covalent?
Ionic bonds usually occur between metal and nonmetal ions. For example, sodium (Na), a metal, and chloride (Cl), a nonmetal, form an ionic bond to make NaCl. In a covalent bond, the atoms bond by sharing electrons.
Is boron a metal?
Boron, the lightest of these elements, is a metalloid. Aluminum, gallium, indium, and thallium are silvery white metals.
What is metallic bonding quizlet?
A metallic bond is the force of attraction between a positively charged metal ion and the valence electrons it shares with other ions of the metal. The electrons move freely around the positive ions, which form a lattice-like structure.
Is barium a metallic bond?
BariumFirst isolationHumphry Davy (1808)Main isotopes of barium
Is carbon a metallic lattice?
Metals, alloys, and salts exist as lattices, as do some types of non-metallic compounds, including silicon oxides and the graphite and diamond forms of carbon. Lattices comprise repeating units, the smallest of which is called a unit cell.
What kind of bond is an alloy?
Metallic bonding is very strong, so the atoms are reluctant to break apart into a liquid or gas. Metallic bonds can occur between different elements. A mixture of two or more metals is called an alloy.
Is lead a metallic bond?
Lead is a relatively unreactive post-transition metal. Its weak metallic character is illustrated by its amphoteric nature; lead and lead oxides react with acids and bases, and it tends to form covalent bonds.
What are alloys examples?
An alloy is a mixture or metallic-solid solution composed of two or more elements. Examples of alloys include materials such as brass, pewter, phosphor bronze, amalgam, and steel.
Is alloy a chemical bond?
Although an alloy can sometimes be a compound (the elements it’s made from are chemically bonded together), it’s usually a solid solution (atoms of the elements are simply intermixed, like salt mixed with water). … Instead of orbiting their atoms, they form a “sea of electrons” surrounding the positive metal ions.
What bonds do nonmetals form?
Nonmetals can form different types of bonds depending on their partner atoms. Ionic bonds form when a nonmetal and a metal exchange electrons, while covalent bonds form when electrons are shared between two nonmetals.
Are alloys ionic or covalent?
In alloys, consumable electrodes are collectivized as in pure metals. However, ionic or covalent chemical bonds appear in alloys because of the presence of two or more kinds of metallic or nonmetallic atoms, between which different chemical interactions are possible.
Is sodium a metallic lattice?
The units occupying lattice points in Sodium, Magnesium, Aluminium are positive ions of them respectively and are surrounded by mobile electrons. Thus Sodium, Magnesium, and Aluminium are metallic solids.
Is silicon a metal?
silicon (Si), a nonmetallic chemical element in the carbon family (Group 14 [IVa] of the periodic table). Silicon makes up 27.7 percent of Earth’s crust; it is the second most abundant element in the crust, being surpassed only by oxygen.
Is Fe a metallic solid?
Metallic Solids. Metallic solids such as crystals of copper, aluminum, and iron are formed by metal atoms Figure 11.8.
Is magnesium a metallic solid?
Elemental magnesium is a gray-white lightweight metal, two-thirds the density of aluminium. Magnesium has the lowest melting (923 K (1,202 °F)) and the lowest boiling point 1,363 K (1,994 °F) of all the alkaline earth metals.
What type of bond is Diamond?
Structure and bonding Diamond is a giant covalent structure in which: each carbon atom is joined to four other carbon atoms by strong covalent bonds.
Is copper a metal or nonmetal?
copper (Cu), chemical element, a reddish, extremely ductile metal of Group 11 (Ib) of the periodic table that is an unusually good conductor of electricity and heat. Copper is found in the free metallic state in nature. This native copper was first used (c.
Is copper heavy or light?
The earliest known metals—common metals such as iron, copper, and tin, and precious metals such as silver, gold, and platinum—are heavy metals. From 1809 onward, light metals, such as magnesium, aluminium, and titanium, were discovered, as well as less well-known heavy metals including gallium, thallium, and hafnium.
Is Cl A metal?
Chlorine is a non-metal. … Chlorine usually accepts electrons so is an oxidant, thus it shows the property of a non-metal. The element chlorine is a yellowish-green gas which is extremely reactive and react with various metals.