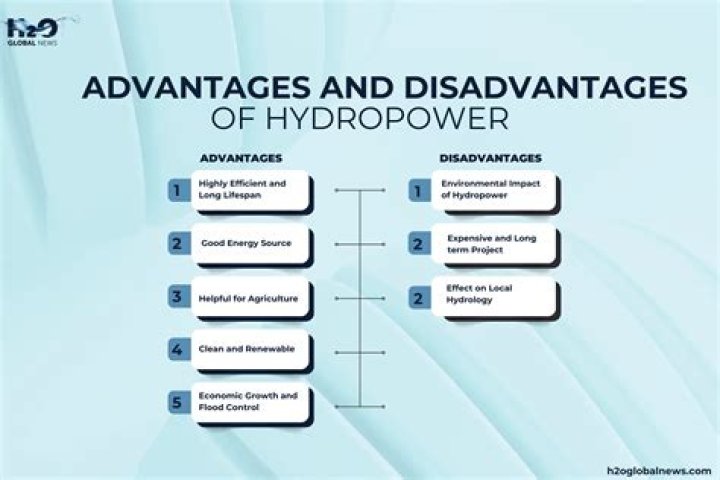
What happens when insulin binds to its receptor
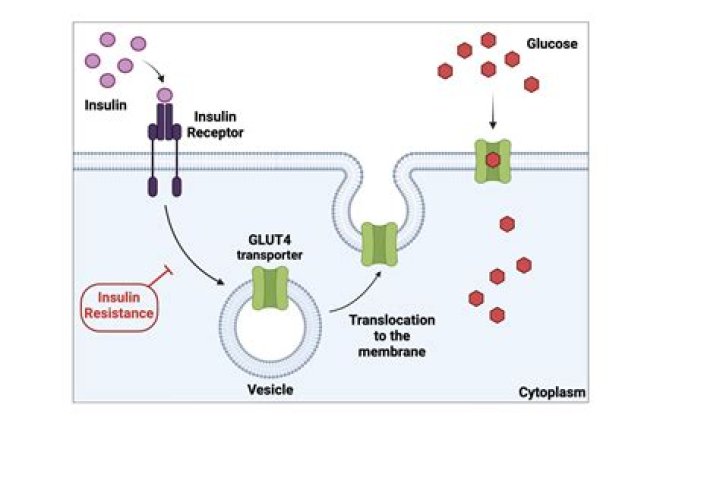
When insulin binds to the cell’s receptor, it results in negative feedback by limiting or stopping some other actions in the cell. It inhibits the release and production of glucose from the cells which is an important part in reducing the glucose blood level.
What happens when insulin binds to insulin receptors?
At the cellular level, insulin binds to the insulin receptor (IR) on the plasma membrane (PM) and triggers the activation of signaling cascades to regulate metabolism and cell growth.
How does insulin binds to cells and the mechanism?
Like a key fits into a lock, insulin binds to receptors on the cell’s surface, causing GLUT4 molecules to come to the cell’s surface. … The vesicles transported GLUT4 to the cell membrane and merged with the membrane, a process known as fusion.
Which process is a major effect of insulin binding to its receptor?
A major effect of insulin acting through its membrane receptor is to stimulate the translocation of GLUT4-containing vesicles in the intracellular cytosol to migrate to the plasma membrane and by the process of exocytosis. Thus the GLUT4 becomes a functional gated channel in the cell membrane.How does insulin bind to cells step by step?
When blood glucose levels rise, insulin from the pancreas travels through the blood stream to a fat cell. Insulin then binds to an Insulin Receptor (IR) found in the cell’s plasma membrane. Phosphate groups are then added to the IR through the process of autophosphorylation.
How does the binding of insulin to its receptor stimulate glucose uptake from the bloodstream?
How does binding of insulin to its receptor cause glucose uptake from the bloodstream? It causes insertion of GLUT4 transporters in the membrane. … The pancreas releases glucagon, which eventually causes blood glucose levels to increase.
What is the action triggered by the binding of insulin and tyrosine kinase?
Insulin activates the insulin receptor tyrosine kinase (IR), which phosphorylates and recruits different substrate adaptors such as the IRS family of proteins. … Insulin stimulates glucose uptake in muscle and adipocytes via translocation of GLUT4 vesicles to the plasma membrane.
Does insulin bind to tyrosine receptors?
In this case, it is the SH2 domain of PI-3 kinase (PI-3K) that binds the P-Tyr of IRS-1. Now activated, PI-3K can convert the membrane lipid phosphatidylinositol 4,5-bisphosphate (PIP2) to phosphatidylinositol 3,4,5-triphosphate (PIP3). This indirectly activates a protein kinase, PKB (Akt), via phosphorylation.What is the function of insulin receptor?
The main physiological role of the insulin receptor appears to be metabolic regulation, whereas all other receptor tyrosine kinases are engaged in regulating cell growth and/or differentiation.
How does insulin trigger the uptake of glucose?Insulin increases glucose uptake mainly by enriching the concentration of Glut4 proteins at the plasma membrane, rather than by increasing the intrinsic activity of the transporter (2,3).
Article first time published onWhat stimulates insulin release?
Insulin secretion is governed by the interaction of nutrients, hormones, and the autonomic nervous system. Glucose, as well as certain other sugars metabolized by islets, stimulates insulin release.
What happens if insulin receptors stop working?
Without insulin, cells are unable to use glucose as fuel and they will start malfunctioning. Extra glucose that is not used by the cells will be converted and stored as fat so it can be used to provide energy when glucose levels are too low.
What protein does insulin bind to?
Insulin receptor uses IRS proteins to activate PI3-kinase. Activated insulin receptor binds transiently IRS proteins, which becomes phosphorylated and thereafter bind the regulatory subunits (p85) of PI3-kinase.
How does insulin alter glucose transport by facilitated diffusion?
Two important effects are: 1. Insulin facilitates entry of glucose into muscle, adipose and several other tissues. The only mechanism by which cells can take up glucose is by facilitated diffusion through a family of hexose transporters.
How is insulin transported?
Insulin is transported into the CNS by a saturable receptor-mediated process that is proposed to be dependent on the insulin receptor. Transport of insulin into the brain is dependent on numerous factors including diet, glycemia, a diabetic state and notably, obesity.
What happens when insulin is secreted?
There are many effects of insulin secretion, including increased glycogen synthesis; increased lipid synthesis; increased esterification of fatty acids; decreased proteolysis; decreased lipolysis; decreased glucogenesis; decreased autophagy; increased amino acid uptake; increased potassium uptake; arterial muscle tone; …
What inhibits the release of insulin?
Several agonists including norepinephrine, somatostatin, galanin, and prostaglandins inhibit insulin release. The inhibition is sensitive to pertussis toxin, indicating the involvement of heterotrimeric Gi and/or Go proteins.
What happens when insulin levels are high?
Because of the largely unrestricted insulin signaling, hyperinsulinemia increases the risk of obesity, type 2 diabetes, and cardiovascular disease and decreases health span and life expectancy. In epidemiological studies, high-dose insulin therapy is associated with an increased risk of cardiovascular disease.
How does insulin enhance the diffusion of glucose across muscle cell membrane?
In skeletal muscle and adipose tissue, insulin promotes membrane trafficking of the glucose transporter GLUT4 from GLUT4 storage vesicles to the plasma membrane, thereby facilitating the uptake of glucose from the circulation.
How is insulin metabolized?
At a cellular level in most tissues, insulin degradation is initiated by the hormone binding to specific receptors. The hormone-receptor complex is processed, including internalization and degradation of at least some of the hormone-receptor complexes.
How does insulin inhibit gluconeogenesis?
A significant mechanism through which insulin is able to modulate STAT3 activity is its hypothalamic action. Insulin action in the hypothalamus stimulates IL-6 production in the liver, and IL-6 in turn suppresses gluconeogenesis by activating STAT3.