What does a high surface tension do to the number of liquid molecules that can stay together
What does the high surface tension do to the number of liquid molecules that can stay together? It keeps the liquid molecules together, The higher the surface tension the more molecules stay on. The water had the highest surface tension and the soapy water had the lowest surface tension.
What happens when surface tension is high?
Surface Tension: “The property of the surface of a liquid that allows it to resist an external force, due to the cohesive nature of its molecules.” … The high surface tension helps the paper clip – with much higher density – float on the water.
What is the liquid with highest surface tension?
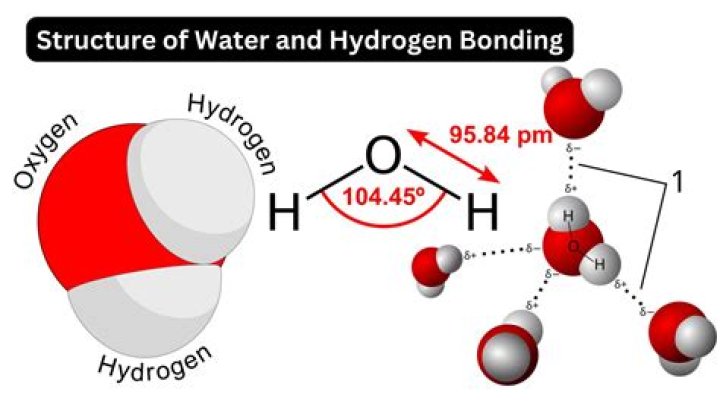
Besides mercury, water has the highest surface tension for all liquids, which is due to the hydrogen bonding in water molecules.
How does surface tension affect intermolecular forces?
The stronger the intermolecular interactions, the greater the surface tension. … It results when cohesive forces, the intermolecular forces in the liquid, are weaker than adhesive forces, the attraction between a liquid and the surface of the capillary.What is the relation between the liquid type and the surface tension?
At liquid–air interfaces, surface tension results from the greater attraction of liquid molecules to each other (due to cohesion) than to the molecules in the air (due to adhesion). … One is an inward force on the surface molecules causing the liquid to contract.
What is the effect of surface tension on number of drops?
The experimental results show that a lower surface tension will result in a higher tangential spread factor and a lower receding rate during the receding stage, for the drop impinging and depositing on a rotating disk. In addition, a lower surface tension of the drop tends to promote the occurrence of splash.
What affects surface tension?
Surface tension is caused by the effects of intermolecular forces at the interface. Surface tension depends on the nature of the liquid, the surrounding environment and temperature. Liquids were molecules have large attractive intermolecular forces will have a large surface tension.
Why does water have a high surface tension intermolecular forces?
The high surface tension of water is caused by strong molecular interactions. The surface tension arises due to cohesive interactions between the molecules in the liquid. At the bulk of the liquid, the molecules have neighboring molecules on each side. … The stronger the cohesive force, the stronger the surface tension.Does high surface tension have strong intermolecular forces?
The surface tension of a liquid is a measure of the elastic force in the liquid’s surface. Liquids with strong intermolecular forces have higher surface tensions than liquids with weaker forces.
Why do polar Liquids have greater surface tension?The molecules below the surface of a liquid are attracted to the molecules all around them. The molecules at the surface do not have other molecules above them, so they are attracted more strongly to their neighbours on the surface. … Water is a polar molecule with strong hydrogen bonds. Its surface tension is 73 mN/m.
Article first time published onWhat liquids have the strongest surface tension oil or water Why?
Water (H2O) due to presence of Hydrogen bonding. So relatively high attraction between water molecules the surface tension is high. Originally Answered: What liquid has the highest surface tension? Mercury, closely followed by water.
Why do substances with high surface tensions also tend to have high viscosities?
3. Why do substances with high surface tension also tend to have high viscosities? Liquids with stronger intermolecular forces of attraction hold the molecules closer together, which causes stronger surface tension and greater resistance to flowing (viscosity).
Does high surface tension mean high viscosity?
If you relate the water and syrup demonstration you will see that a higher viscosity means a greater surface tension and a lower viscosity means a lower surface tension.
Why does water have the highest surface tension with lowest viscosity?
Water’s high surface tension is due to the hydrogen bonding in water molecules. … Water has very strong intermolecular forces, hence the low vapor pressure, but it’s even lower compared to larger molecules with low vapor pressures. Viscosity is the property of fluid having high resistance to flow.
What does surface tension and surface energy mean?
The attractive force of the molecules present at the surface of a liquid towards each other is called the surface tension of that liquid. Surface energy is the equivalent attractive force present between the molecules at the surface of a solid substance.
Why does surface tension of a liquid increases with decreasing temperature?
The strength of surface tension depends on intermolecular forces. As temperature increases, molecules of liquid become more active and they move more rapidly; therefore, the intermolecular forces are more instable. Surface tension decreases with increasing temperature.
Do all liquids have surface tension?
No, liquids have different surface tensions. Partly due to the strength of the force that bids the molecules together. Many liquids have what is called hydrogen bonding and water has a high value for hydrogen bonding.
What is the role of surface tension in everyday life?
The effects of surface tension are of central importance in many everyday phenomena: it causes small droplets of rain to stick to your windows, creates bubbles when you add detergent in your sink, and propels water-striding insects on the surface of ponds.
Why surface tension decreases with increase in concentration?
The surface tension decreases with increase in concentration. This is due to breaking of hydrogen bonds when surfactants are added in water, leading to higher adsorption at the air-water interface. … With increase in temperature the surface tension decreased steadily.
What is the effect on surface tension of temperature?
Surface tension decreases with an increase in temperature. So these are the effects of temperature on viscosity and surface tension. In general, surface tension decreases when the temperature increases because cohesive forces decrease with increased molecular thermal activity.
What is surface tension What is its cause why is the surface tension also called surface energy?
Surface tension is the energy, or work, required to increase the surface area of a liquid due to intermolecular forces. Since these intermolecular forces vary depending on the nature of the liquid (e.g. water vs. … Water droplets form on the surface of the water pool because of surface tension.
How do intermolecular forces affect the surface tension and viscosity as intermolecular forces increase what happens to each property?
As the intermolecular attraction increases, • The vapor pressure ( the pressure of the vapor that is in equilibrium with its liquid) decreases • The boiling point ( the temperature at which the vapor pressure becomes equal to the pressure exerted on the surface of the liquid) increases • Surface tension ( the …
How does the viscosity and surface tension of liquids change as intermolecular forces become stronger?
Viscosity is the resistance of liquids to flow. Viscosity increases with 1) stronger intermolecular forces and 2) decreasing temperature. Surface tension results from the net inward force experienced by the molecules on the surface of a liquid.
What indicates strong intermolecular forces in a liquid?
Melting Point – The temperature at which a solid turns into a liquid. High melting points indicate STRONG intermolecular forces.
How does the surface tension of water compare to the surface tension of other liquids?
How does the surface tension of water compare with the surface tensions of most other liquids? It is higher.
Is high surface tension applicable for polar molecule?
Surface tension is generally expressed in N/m (SI unit). Typically, polar solvents have higher surface tension than their non-polar counterparts.
How does polarity affect surface tension?
When a polar substance is put in water, the positive ends of its molecules are attracted to the negative ends of the water molecules, and vice versa. … Because of their polarity, water molecules are strongly attracted to one another, which gives water a high surface tension.
Why does water have a higher surface tension than alcohol?
Water has greater degrees of hydrogen-bonding in the bulk liquid. … As a result, it is more difficult to deform the surface of water than the surface of ethyl alcohol. Therefore, since water molecules on a liquid surface are harder to push down on the surface tension is higher for water than for ethyl alcohol.
What happens to the surface tension of water on adding detergent to it?
When detergent is added to water, it decreases the surface tension of the water. … As the detergent spreads through the water, it decreases the surface tension throughout the water, and the raft stops moving eventually because there is no longer a difference in the surface tension.
Is surface tension due to cohesive or adhesive forces or both?
Attractive forces between molecules of different types are called adhesive forces. Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. This general effect is called surface tension.
How does surface tension affect viscosity?
Surprisingly, we found that the solutions with a higher viscosity than water had either less or the same surface tension as water, and we suspect this is due to the unchanged intermolecular bonding of water molecules (hydrogen bonding) causing surface tension as viscosity increased.