What did Rutherfords gold foil experiment suggest about the structure of an atom
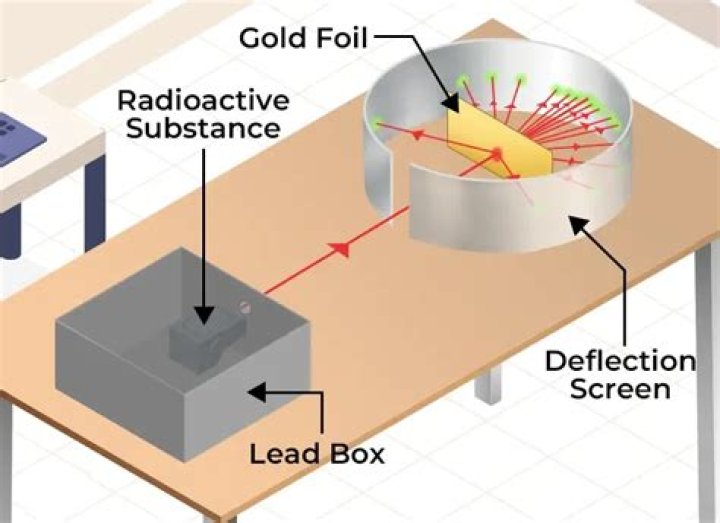
Rutherford’s gold foil experiment showed that the atom is mostly empty space with a tiny, dense, positively-charged nucleus. Based on these results, Rutherford proposed the nuclear model of the atom.
What did Rutherford's gold-foil experiment suggest about the structure of an atom quizlet?
Ernest Rutherford experiment used particles and reflection to determine the structure of the atom. … He concluded that there must be something inside an atom for the alpha particles to bounce off, that must be small, dense, and positively charged.
What did Rutherford conclude by using his gold-foil experiment?
Physicist Ernest Rutherford established the nuclear theory of the atom with his gold-foil experiment. When he shot a beam of alpha particles at a sheet of gold foil, a few of the particles were deflected. He concluded that a tiny, dense nucleus was causing the deflections.
What did Rutherford conclude about the structure of the atom?
He concluded that all of the positive charge and the majority of the mass of the atom must be concentrated in a very small space in the atom’s interior, which he called the nucleus. The nucleus is the tiny, dense, central core of the atom and is composed of protons and neutrons.What did the gold foil experiment prove quizlet?
What were the conclusions of the Goldfoil Experiment? Subatomic particles with no charge but with a mass nearly equal to that of a proton. Who discovered neutrons? The tiny central core of an atom and is composed of protons and neutrons.
What did Rutherford conclude about the structure of the atom quizlet?
What did Rutherford conclude about the structure of an atom? … The total number of protons and neutrons in thr nucleus of an atom.
Why was the gold foil experiment so important?
The gold foil experiment showed that the atom has a positively charged nucleus which holds most of its mass. It was the basis for the nulear model of the atom.
What were the conclusions of Rutherford in his experiment?
Conclusion of Rutherford’s scattering experiment: Most of the space inside the atom is empty because most of the α-particles passed through the gold foil without getting deflected. Very few particles were deflected from their path, indicating that the positive charge of the atom occupies very little space.What did Rutherford conclude about the structure of an atom from his experiment of scattering of alpha particles?
From the α-particle scattering experiment, Rutherford concluded that. α-particles can come within a distance of the order of 10−15m of the nucleus.
What three conclusions came from the gold foil experiment?Thus the conclusions made were: Atom has a very small nucleus at the centre. There is large empty space around the nucleus. Entire mass of an atom is concentrated in a very small positively charged region which is called the nucleus. Electrons are distributed in the vacant space around the nucleus.
Article first time published onWhat did Rutherford do quizlet?
Rutherford had found the positively charged nucleus in the middle of every atom using his Gold Foil Experiment. While doing this experiment, he expected these particles to just pass right through the foil but they bounced right back. He also proposed there were negatively charged electrons revolving around the nucleus.
What was the most important finding in Rutherford's gold foil experiment quizlet?
Most alpha particles in Rutherford’s experiment passed straight through a sheet of gold foil without being deflected. What discovery indicated that atoms are mostly empty space? The particles of the ray were SMALLER than atoms and were NEGATIVELY charged.
What describes Rutherford's experiment?
Which describes Ernest Rutherford’s experiment? Positive particles are fired at gold foil. Most pass through, but some bounce back. a cation that has a smaller radius than the atom.
How did scientist prove the structure of an atom?
The Rutherford Gold Foil Experiment offered the first experimental evidence that led to the discovery of the nucleus of the atom as a small, dense, and positively charged atomic core. … With Geiger and Marsden’s experimental evidence, Rutherford deduced a model of the atom, discovering the atomic nucleus.
What did Rutherford conclude about the internal structure of an atom group answer choices?
Rutherford concluded from his metal foil experiments that most of an atom is empty space with a tiny, dense, positively charged nucleus at the center that contains most of the mass of the atom. He also concluded that the electrons orbit the nucleus like the planets orbit the sun.
What structure did Rutherford discover quizlet?
slightly different types or versions of atoms of the same element. Isotopes are atoms of the same element that have a different number of neutrons. In 1911, Ernest Rutherford and his colleagues discovered the nucleus of the atom using their famous gold foil experiment.
Which of the following conclusions did Rutherford make after conducting his gold foil experiment with alpha particles?
Rutherford found that a small percentage of alpha particles were deflected at large angles, which could be explained by an atom with a very small, dense, positively-charged nucleus at its center (bottom). were deflected by the gold foil.
What did Rutherford conclude from the observation of a ray scattering experiment?
Conclusion of Rutherford’s scattering experiment: Most of the space inside the atom is empty because most of the α-particles passed through the gold foil without getting deflected. Very few particles were deflected from their path, indicating that the positive charge of the atom occupies very little space.
What is gold foil experiment write the conclusions and shortcomings of Rutherford's model of atom?
On the basis of gold foil experiment, Rutherford concluded that an atom consists of nucleus which has positive charge and it is surrounded with electrons which are moving around the nucleus. The number of electrons and protons are equal and the entire mass of the atom is concentrated at its nucleus.
What was Rutherford's experiment quizlet?
A: Rutherford concluded that the positive charges of an atom must be only in the middle, and that the negatively charged electrons are on the outside of the atoms. Also that the closer the ray got to the positively charged center more drastic deflections occurred.
How did Rutherford discover the proton?
In 1909, Rutherford discovered proton in his famous gold foil experiment. He bombarded alpha particles on an ultrathin gold foil. … Based on Wilhelm Wien’s theory, who in 1898 discovered the proton in streams of ionized gas, Rutherford postulated the hydrogen nucleus to be a new particle in 1920, which he called proton.
What is gold foil experiment Class 9?
Rutherford in 1911, carried out an experiment called ‘Gold foil experiment’ and could conclude the nature of an atom and the position of the protons present in the atom decisively. He also proposed the position and behaviour of electrons. They bombarded fragile sheets of gold foil with fast-moving alpha particles.
Which experiments was key along with Rutherford experiment to understand the composition and structure of atoms?
Distributed on the atom volume. Which of the following experiments was key, along with Rutherford experiment to understand the composition and structure of atoms. Thomson cathode ray experiment.
How did Rutherford's gold foil experiment show that the atomic nucleus is both small and very massive?
In what way did Rutherford’s gold-foiling scattering experiment show that the atomic nucleus is both small and very massive? … The small number of deflected particles were repelled from powerful heavy positively charged central cores which is known as the atomic nucleus.
How did Rutherford experiment proved the existence of the nucleus?
Rutherford’s experiment showed the existence of a nuclear atom – a small, positively-charged nucleus surrounded by empty space and then a layer of electrons to form the outside of the atom. Most of the alpha particles did pass straight through the foil.
Why it is important to understand the structure of an atom?
As a result of the work done by previous scientists on atomic models, scientists now have a good idea of what an atom looks like. This knowledge is important because it helps us to understand why materials have different properties and why some materials bond with others.
How did the structure of atom develop?
In 1911, Ernest Rutherford developed the first coherent explanation of the structure of an atom. Using alpha particles emitted by radioactive atoms, he showed that the atom consists of a central, positively charged core, the nucleus, and negatively charged particles called electrons that orbit the nucleus.