What are the subshells of an atom
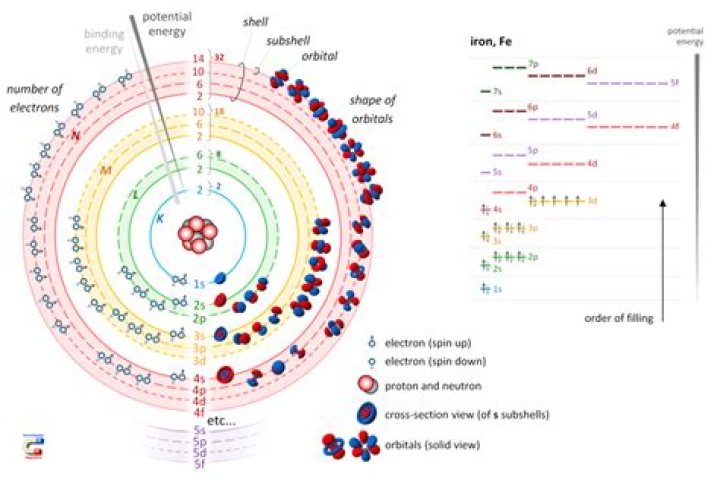
Within the shells, electrons are further grouped into subshells of four different types, identified as s, p, d, and f in order of increasing energy. The first shell has only an s subshell; the second shell has an s and a p subshell; the third shell has s, p, and d subshells, and the fourth has s, p, d and f subshells.
What is the order of Subshells in an atom?
This gives the following order for filling the orbitals: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p, (8s, 5g, 6f, 7d, 8p, and 9s)
What are the Subshells called?
There are 4 subshells, s, p, d, and f. Each subshell can hold a different number of electrons. The n number determines how many of the subshells make up the shell.
What are in Subshells?
A subshell is a group of orbitals. … Orbitals each hold two electrons, and electrons in an orbital with the same principle quantum number, angular momentum quantum number, and magnetic quantum number, ml are part of the same orbital. The ml is what distinguishes different orbitals in a subshell.How do you find Subshells?
Subshells are determined by the value of l; thus, we first determine which values of l are allowed, and then we apply the equation “maximum number of electrons that can be in a subshell = 2(2l + 1)” to find the number of electrons in each subshell.
What does 1s 2s 2p mean?
The superscript is the number of electrons in the level. … The number in front of the energy level indicates relative energy. For example, 1s is lower energy than 2s, which in turn is lower energy than 2p. The number in front of the energy level also indicates its distance from the nucleus.
What is 1s 2s 2p?
1s will be filled first, with the maximum of 2 electrons. • 2s will be filled next, with the maximum of 2 electrons. • 2p will be filled next, with the maximum of 6 electrons.
How are atom shells filled?
Energy shellMaximum number of electronsFirst2Second8Third8Whats a subshell?
Definition of subshell : any of the one or more orbitals making up an electron shell of an atom.
How many Subshells are there in K shell?The first K shell has one subshell, called ‘1s’; the L shell has two subshells, called ‘2s’ and ‘2p’; the third shell has ‘3s’, ‘3p’, and ‘3d’; and so on.
Article first time published onHow do you write SPDF?
There are different orbital shapes (s,p,d,f) Each orbital can only hold 2 electrons max. There is a hierarchy, i.e. s orbitals will be filled before p orbitals which will be filled before d orbitals and so on. (s<p<d<f) (note, this is a general rule but there are exceptions)
How many orbitals are in each subshell?
This number divides the subshell into individual orbitals which hold the electrons; there are 2l+1 orbitals in each subshell. Thus the s subshell has only one orbital, the p subshell has three orbitals, and so on.
How many electrons are in 2p?
The second shell has two subshells, s and p, which fill with electrons in that order. The 2s subshell holds a maximum of 2 electrons, and the 2p subshell holds a maximum of 6 electrons.
How many Subshells are associated?
The four sub-shells are associated with n = 4, which are s, p, d and f. The number of orbitals = 16.
How do you find subshells and orbitals?
The number of orbitals in a shell is the square of the principal quantum number: 12 = 1, 22 = 4, 32 = 9. There is one orbital in an s subshell (l = 0), three orbitals in a p subshell (l = 1), and five orbitals in a d subshell (l = 2). The number of orbitals in a subshell is therefore 2(l) + 1.
What are the subshells of orbitals that hold the electrons?
Principal shell 3n has s, p, and d subshells and can hold 18 electrons. Principal shell 4n has s, p, d, and f orbitals and can hold 32 electrons. Moving away from the nucleus, the number of electrons and orbitals found in the energy levels increases.
Does the 3d sublevel exist?
5. Does the 3f sublevel exist? (Note: the “3” stands for the 3rd energy level.) No, in the 3rd energy level there are only s, p, and d sublevels. The following sublevels exist in the 3rd energy level: 3s, 3p, and 3d.
Is 2d a possible sublevel?
In the 2nd energy level, electrons are placed only in the ‘s’ and ‘p’ sublevels, so there is no ‘d’ orbital. i.e. The second principal energy level consists of two sub-shell which is 2s (ℓ=0) and 2p (ℓ=1). So, 2d orbital can’t exist.
What is e9 class configuration?
Electronic configuration is defined as the distribution of electrons into the orbitals of an atom. Every neutral atom consists of a fixed number of electrons which is equal to the number of protons and is called the atomic number.
What is SPDF rule?
There are different orbital shapes (s,p,d,f) Each orbital can only hold 2 electrons max. There is a hierarchy, i.e. s orbitals will be filled before p orbitals which will be filled before d orbitals and so on. (s<p<d<f) (note, this is a general rule but there are exceptions)
What is SPDF in periodic table?
From Wikipedia, the free encyclopedia. Spdf or SPDF may refer to: Electron configuration, for which there is an obsolete system of categorizing spectral lines as “sharp”, “principal”, “diffuse” and “fundamental”; also the names of the sub shells or orbitals. The blocks of the periodic table.
What is SPDF block elements?
Elements in the long form of periodic table have been divided into four blocks i.e. s ,p ,d and f. This division is based upon the name of the orbitals which receives the last electron. … The last electron in these elements also enter the s orbital i.e. 1s, 2s, 3s, 4s, 5s, 6s and 7s orbital.
What is inside an electron?
Right now, our best evidence says that there are particles inside of neutrons and protons. Scientists call these particles quarks. Our best evidence also shows us that there is nothing inside of an electron except the electron itself.
How many Subshells are there in fourth shell?
Re: How many subshells there are in the shell n=4? Answer: There are 4 subshells: 4s, 4p, 4d, and 4f.
What is the difference between subshells and orbitals?
A subshell is composed of orbitals. It is a subdivision of electron shells which is separated by electron orbitals. … Each subshell contains one or more orbitals. s contain one orbital, p contains 3 orbitals, d contains 5 orbitals and f contain 7 orbitals.
What order do Subshells fill?
The order of filling subshells is the same: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, etc.
Which two particles are the same number in an atom?
An atom contains equal numbers of protons and electrons .
What are the D block elements called?
The d-block elements are found in groups 3, 4, 5, 6, 7, 8, 9, 10, 11, and 12 of the periodic table; d-block elements are also known as the transition metals. The d orbital is filled with the electronic shell “n-1.”
What is the M shell?
Definition of M-shell : the third innermost shell of electrons surrounding an atomic nucleus — compare k-shell , l-shell.
Which shell is incomplete in calcium?
So, the shells incomplete in the Ca atom are M and N shells as M shell can contain up to 18 electrons and N shell can have 8 electrons.
Which Subshell is common to all shells?
So,the s subshell is there for every shell. It is the common one.