What are the properties and behavior of gases
The Properties of Gases. Gases have three characteristic properties: (1) they are easy to compress, (2) they expand to fill their containers, and (3) they occupy far more space than the liquids or solids from which they form.
What are the behavior of gases?
The behavior of gases can be modeled with gas laws. Boyle’s law relates a gas’s pressure and volume at constant temperature and amount. Charles’s law relates a gas’s volume and temperature at constant pressure and amount. In gas laws, temperatures must always be expressed in kelvins.
What are the 4 properties of gases?
Because most gases are difficult to observe directly, they are described through the use of four physical properties or macroscopic characteristics: pressure, volume, number of particles (chemists group them by moles) and temperature.
What are the 5 properties of gases?
- Low Density. Gases contain scattered molecules that are dispersed across a given volume and are therefore less dense than in their solid or liquid states. …
- Indefinite Shape or Volume. Gases have no definite shape or volume. …
- Compressibility and Expandability. …
- Diffusivity. …
- Pressure.
What are the 7 properties of gases?
- What are the Properties of Gases? Gasses do not possess any definite volume or shape. …
- Compressibility. Particles of gas have huge intermolecular spaces in the midst of them. …
- Expansibility. When pressure is exerted on gas, it contracts. …
- Diffusibility. …
- Low Density. …
- Exertion of Pressure.
Why is it important to know the behavior of gases?
Introduction. The study of gases allows us to understand the behavior of matter at its simplest: individual particles, acting independently, almost completely uncomplicated by interactions and interferences between each other.
What are the 4 factors that affect the behavior of gases?
Four variables are used to describe the condition of a gas. They are pressure (P), volume (V), temperature (T), and the amount of the gas as measured by the number moles (n).
What are the properties of gases Class 9?
- Gas has indefinite shape.
- Gas has no fixed volume.
- Gas gets the shape and volume of container.
- Gas fills the container completely.
- Gas has very low density.
- Because of low density gas are light.
- Gas can flow easily and hence are called fluid.
What are properties of gases?
The Properties of Gases. Gases have three characteristic properties: (1) they are easy to compress, (2) they expand to fill their containers, and (3) they occupy far more space than the liquids or solids from which they form.
Which of the following are characteristics of gases?- no definite shape (takes the shape of its container)
- no definite volume.
- particles move in random motion with little or no attraction to each other.
- highly compressible.
What are the properties of gases quizlet?
–Gas particles have no volume. – Particles move in constant, straight line, random motion. -Collisions are elastic. -There are no forces of attraction or repulsion between molecules.
Can the gas laws be explained by molecular behavior?
The behavior of ideal gases is explained by the kinetic molecular theory of gases. Molecular motion, which leads to collisions between molecules and the container walls, explains pressure, and the large intermolecular distances in gases explain their high compressibility.
What is not a property of gases?
Gases have a specific shape or volume. There is no fixed volume or shape in the gaseous state. It means that there is no structure in gas. The gaseous pressure is caused by countless collisions of molecules on the container walls.
What are the 4 gas laws?
Gas Laws: Boyle’s Law, Charle’s Law, Gay-Lussac’s Law, Avogadro’s Law.
What are the 3 factors that describe gases?
This third factor is part of the ideal gas law, which explains how these three factors — temperature, volume and density — interact with each other.
What are the 4 physical measurable characteristics of a gas?
To describe a gas fully, you need to state four measurable quantities: volume, temperature, number of molecules, and pressure. You already know what is meant by volume, temperature, and number of molecules. In this section, you will learn about pressure and its measurement.
What are the properties of gases upon describing the gas laws?
The gas laws describe the relationships between a gas’s temperature, pressure, volume, and amount.
What are 3 examples of a gas?
- Hydrogen.
- Nitrogen.
- Oxygen.
- Carbon Dioxide.
- Carbon Monoxide.
- Water Vapour.
- Helium.
- Neon.
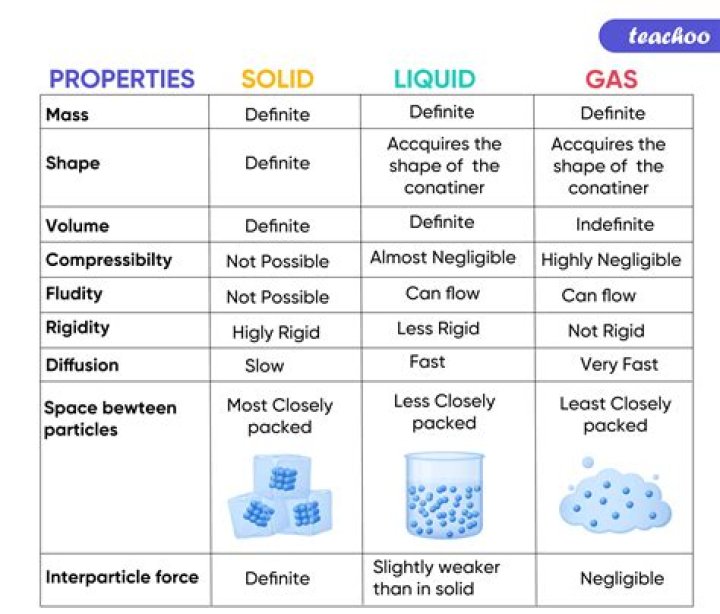
What are the three properties of solid liquid and gas?
- Solids – relatively rigid, definite volume and shape. In a solid, the atoms and molecules are attached to each other. …
- Liquids – definite volume but able to change shape by flowing. In a liquid, the atoms and molecules are loosely bonded. …
- Gases – no definite volume or shape.
What are the 5 properties of liquid?
- Liquids are almost incompressible. In liquids molecules are pretty close to each other. …
- Liquids have fixed volume but no fixed shape. …
- Liquids flow from higher to lower level.
- Liquids have their boiling points above room temperature, under normal conditions.
What property indicates whether the gas is hot or cold?
The temperature of a gas is a measure of the average translational kinetic energy of the molecules. In a hot gas, the molecules move faster than in a cold gas; the mass remains the same, but the kinetic energy, and hence the temperature, is greater because of the increased velocity of the molecules.
What can gases do?
They vibrate and move freely at high speeds. A gas will fill any container, but if the container is not sealed, the gas will escape. Gas can be compressed much more easily than a liquid or solid.
Which property of gases is explained by the assumption that gases are mostly empty space?
There is no space between the individual particles, so they cannot pack together. The kinetic-molecular theory explains why gases are more compressible than either liquids or solids. Gases are compressible because most of the volume of a gas is composed of the large amounts of empty space between the gas particles.
How do gases turn into solids?
Under certain circumstances, gas can transform directly into a solid. This process is called deposition. Water vapor to ice – Water vapor transforms directly into ice without becoming a liquid, a process that often occurs on windows during the winter months.
Which of these describe the behavior of an ideal gas?
For a gas to be “ideal” there are four governing assumptions: The gas particles have negligible volume. The gas particles are equally sized and do not have intermolecular forces (attraction or repulsion) with other gas particles. … The gas particles have perfect elastic collisions with no energy loss.
How do you explain the behavior of gases in terms of the kinetic molecular theory?
Kinetic Molecular Theory states that gas particles are in constant motion and exhibit perfectly elastic collisions. Kinetic Molecular Theory can be used to explain both Charles’ and Boyle’s Laws. The average kinetic energy of a collection of gas particles is directly proportional to absolute temperature only.
What are fundamental concepts that are needed to model the Behaviour of gases?
The three fundamental gas laws discover the relationship of pressure, temperature, volume and amount of gas. Boyle’s Law tells us that the volume of gas increases as the pressure decreases. Charles’ Law tells us that the volume of gas increases as the temperature increases.
Which of the following is not a property of air?
(d) it is a compound is not the property of air. Air is a combination of gases and hence it is a mixture. Compound is characterized by chemical bonding between the elements. Mixture is formed by physical mixing of elements which is seen in air.
Which of the following properties of gas is definite in an ideal gas?
The properties of IDEAL gases are that: they are compressible. they are always in motion, colliding elastically within a container. they take the shape of the container.
Does gas have mass?
Gases have mass. The space between gas particles is empty. Gases can be formed as products in chemical reactions. Gas particles can form bonds between them under certain conditions.
What are gas laws?
gas laws, laws that relate the pressure, volume, and temperature of a gas. … These two laws can be combined to form the ideal gas law, a single generalization of the behaviour of gases known as an equation of state, PV = nRT, where n is the number of gram-moles of a gas and R is called the universal gas constant.