What are the carboxylic acid derivatives
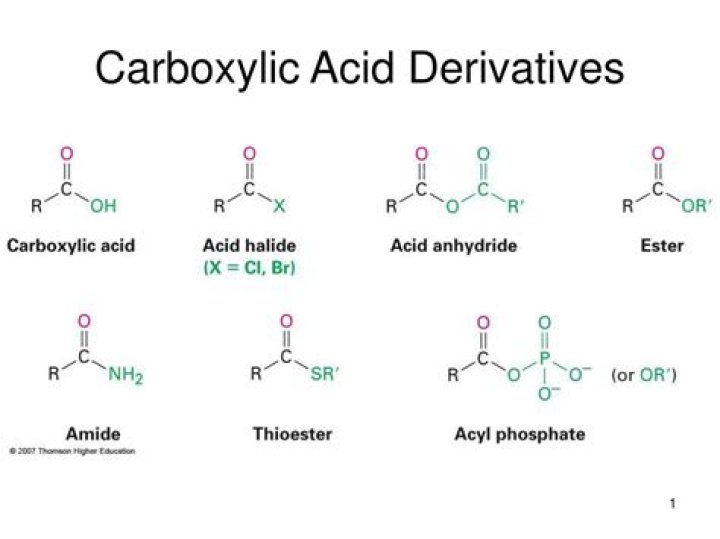
The functional groups at the heart of this chapter are called carboxylic acid derivatives: they include carboxylic acids themselves, carboxylates (deprotonated carboxylic acids), amides, esters, thioesters, and acyl phosphates. Cyclic esters and amides are referred to as lactones and lactams, respectively.
What are the derivatives of carboxylic acid write the names?
FormulaIUPAC NameMolecular WeightCH3CO2CHOethanoic methanoic anhydride88CH3CH2CO2CH3methyl propanoate88CH3CO2C2H5ethyl ethanoate88CH3CH2COClpropanoyl chloride92.5
How each of the acid derivatives is produced from carboxylic acid?
Carboxylic acids have a hydroxyl group bonded to an acyl group, and their functional derivatives are prepared by replacement of the hydroxyl group with substituents, such as halo, alkoxyl, amino and acyloxy. The carboxylic acid derivatives can all be hydrolyzed to carboxylic acids.
What structures are carboxylic acid derivatives?
- The carboxylic acid derivatives all include an acyl group, an R-group bonded to a carbonyl carbon. …
- Although there are many types of carboxylic acid derivatives known, this chapter focuses on four: acid halides (acyl halides), acid anhydrides, esters, and amides.
What are examples of carboxylic acids?
Some common examples of carboxylic acids include acetic acid (a component of vinegar) and Formic acid.
Is acid anhydride a derivative of carboxylic acid?
Compounds in which the −OH group of the carboxylic acid is replaced by other functional groups are called carboxylic acid derivatives, the most important of which are acyl halides, acid anhydrides, esters, and amides.
What are four derivatives of carboxylic acid?
The functional groups at the heart of this chapter are called carboxylic acid derivatives: they include carboxylic acids themselves, carboxylates (deprotonated carboxylic acids), amides, esters, thioesters, and acyl phosphates.
What are acid derivatives definition?
Carboxylic acid derivative: A carbonyl-containing functional group other than an aldehyde or ketone, or a molecule containing such a functional group. Called a carboxylic acid derivative because these functional groups can, in principle, be produced (derived) from the corresponding carboxylic acid.Is nitrile a derivative of carboxylic acid?
Despite not containing an acyl group, nitriles are still considered carboxylic acid derivatives since on hydrolysis they form carboxylic acids. These are most commonly made via the conversion of a carboxylic acid to a primary amide, followed by dehydration.
Is acid chloride a derivative of carboxylic acid?Carboxylic acids react with phosphorus tribromide to form acid bromides. Acid chlorides are the most reactive carboxylic acid derivative, which allows them to easily convert to other acyl compounds.
Article first time published onWhy are esters derivatives of carboxylic acids?
Esters and amides are considered to be derivatives of carboxylic acids because the OH in the carboxyl group is replaced with another group.
Why are carboxylic acids derivatives important?
Carboxylic acids and their derivatives are used in the production of polymers, biopolymers, coatings, adhesives, and pharmaceutical drugs. They also can be used as solvents, food additives, antimicrobials, and flavorings.
Which is not the derivative of carboxylic acid?
Which of the following is not a derivative of a carboxylic acid? Explanation: Aldehyde is not a derivative of carboxylic acid. Esters can be derived from carboxylic acids by reacting them with to form an acid chloride.
Which molecule is a carboxylic acid?
Carboxylic acidpKaTrifluoroacetic acid (CF3CO2H)0.23
What are the first 5 carboxylic acids?
Carboxylic acids is a homologous series in which the compounds contain a functional group called the carboxyl group (-COOH). … The first four carboxylic acids derived from alkanes are methanoic acid (HCOOH), ethanoic acid (CH3COOH), propanoic acid (C2H5COOH) and butanoic acid (C3H7COOH).
What is another name for carboxylic acid?
FormulaCommon NameIUPAC NameHCO2Hformic acidmethanoic acidCH3CO2Hacetic acidethanoic acidCH3CH2CO2Hpropionic acidpropanoic acidCH3(CH2)2CO2Hbutyric acidbutanoic acid
Which is more reactive carboxylic acid or its derivatives?
Among the carboxylic acid derivatives, carboxylate groups are the least reactive towards nucleophilic acyl substitution, followed by amides, then carboxylic esters and carboxylic acids, thioesters, and finally acyl phosphates, which are the most reactive among the biologically relevant acyl groups.
Is carboxylic acid reactive?
Carboxylic acids and esters are in the middle range of reactivity, while thioesters are somewhat more reactive. The most reactive of the carboxylic acid derivatives frequently found in biomolecules are the acyl phosphates.
What is reaction carboxylic acid?
Carboxylic acids react with Thionyl Chloride (SOCl2) to form acid chlorides. Mechanism. Carboxylic acids can react with alcohols to form esters in a process called Fischer esterification. Basic Reaction. Going from reactants to products simplified.
Is amine an acid derivatives?
Amines are derivatives of ammonia in which one or more of the hydrogen atoms are replaced by an alkyl or aryl group. Depending on the number of R groups (alkyl or aryl group), amines are classified as primary, secondary and tertiary amines. … Amino acids are the building blocks of proteins and peptides.
Which is more reactive ester or carboxylic acid?
Under the reaction condition s the carboxylic acid will deprotonate to give the carboxylate which is a very poor electrophile (after all, it has a negative charge !) so the ester is more reactive than the acid.
What is the characteristic reaction of carboxylic acid derivatives?
Nucleophilic acyl substitutions is the characteristic reaction of carboxylic acid derivatives. This reaction occurs with both negatively charged nucleophiles and neutral nucleophiles. Nucleophilic addition to a carbonyl forms a tetrahedral intermediate with two possible leaving groups, Z or Nu.
What is the molecular formula of carboxylic acid?
The general molecular formula for carboxylic acid is CnH2n+1COOH. Carboxylic acids are nothing but organic compounds in which the carbon atom is bonded with an oxygen atom in the form of a double bond. The carbon atom can also be bonded to a hydroxyl group (−OH) by a single bond.
Are derived from carboxylic acid and an amine?
Amides are produced through the reaction between carboxylic acid and ammonia or amine in the presence of heat.
What are some carboxylic acids in daily life?
Lactic acid, salicylic acid, acetyl salicylic acid and citric acid are examples of carboxylic acids that commonly encounter in everyday life.
What are the sources of alkanoic acid?
The alkanoic acid or carboxylic acids are also called fatty acids because some of them are found in natural fats and oils. They contain the functional group called carboxyl group.
Which of the following is not a carboxylic acid *?
Picric acid is an organic compound with the formula (O2N)3C6H2OH. Its IUPAC name is 2,4,6-trinitrophenol (TNP). It is one of the most acidic phenols (alcohols). Like other highly nitrated organic compounds, picric acid is highly explosive.
How do you name carboxylic acids?
In general, carboxylic acids are named based on the number of carbons in the longest continuous chain, including the carboxyl group (-COOH). The suffix of this carbon chain is then replaced, as carboxylic acids always end in “-oic acid.” An example is CH2O2, in which the longest continuous carbon chain is a methane.
Where are carboxylic acids found?
Many carboxylic acids occur naturally in plants and animals. Citrus fruits such as oranges and lemons contain citric acid. Citric acid is a large carboxylic acid with three ionizable hydrogen atoms. It is found in citrus fruits and gives them their sour or tart flavor.
What are two abbreviations for a carboxylic acid?
AcronymDefinitionCOOHCarboxylic Acid (functional group)



