What are physical and chemical properties of elements
Physical properties include color, density, hardness, and melting and boiling points. A chemical property describes the ability of a substance to undergo a specific chemical change. To identify a chemical property, we look for a chemical change.
What are physical properties of element?
Physical properties include: appearance, texture, color, odor, melting point, boiling point, density, solubility, polarity, and many others.
What are 4 physical properties of elements?
These properties include color, density, melting point, boiling point, and thermal and electrical conductivity. While some of these properties are due chiefly to the electronic structure of the element, others are more closely related to properties of the nucleus, e.g., mass number.
What are the chemical property of an element?
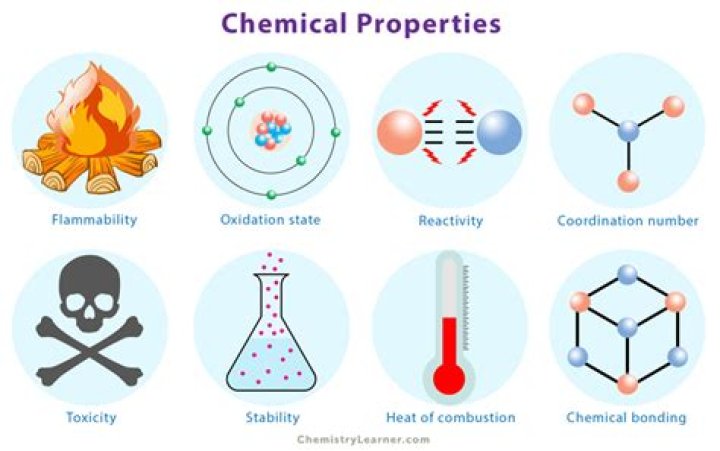
A chemical property is a characteristic of a particular substance that can be observed in a chemical reaction. Some major chemical properties include flammability, toxicity, heat of combustion, pH value, rate of radioactive decay, and chemical stability.What are the 10 physical chemical properties?
- color (intensive)
- density (intensive)
- volume (extensive)
- mass (extensive)
- boiling point (intensive): the temperature at which a substance boils.
- melting point (intensive): the temperature at which a substance melts.
What is a physical property in science?
Definition of physical property : a property (as color, hardness, boiling point) of matter not involving in its manifestation a chemical change.
What are 5 chemical properties examples?
Examples of chemical properties include flammability, toxicity, acidity, reactivity (many types), and heat of combustion.
What are the types chemical properties?
- Toxicity.
- Reactivity.
- Types of chemical bonds formed.
- Coordination number.
- Oxidation states.
- Flammability.
- Heat of combustion.
- Enthalpy of formation.
What are the 5 physical elements?
Everything in nature is made up of five basic elements: earth, water, fire, air, and space. Knowledge of the five elements allows the yogi to understand the laws of nature and to use yoga to attain greater health, power, knowledge, wisdom and happiness.
What are the 15 properties of matter?The properties of matter include any traits that can be measured, such as an object’s density, color, mass, volume, length, malleability, melting point, hardness, odor, temperature, and more.
Article first time published onWhat are 5 chemical and physical changes?
Examples of chemical changes are burning, cooking, rusting, and rotting. Examples of physical changes are boiling, melting, freezing, and shredding. Many physical changes are reversible, if sufficient energy is supplied. The only way to reverse a chemical change is via another chemical reaction.
What are 4 examples of physical properties?
- color (intensive)
- density (intensive)
- volume (extensive)
- mass (extensive)
- boiling point (intensive): the temperature at which a substance boils.
- melting point (intensive): the temperature at which a substance melts.
What is a chemical property in science?
Definition of chemical property : a property of a substance relating to its chemical reactivity (as the explosive property of nitroglycerin)
What is a physical property called?
The physical properties of an object that are traditionally defined by classical mechanics are often called mechanical properties. Other broad categories, commonly cited, are electrical properties, optical properties, thermal properties, etc.
What are the five chemical elements?
Atomic numberName chemical elementSymbol5BoronB6CarbonC7NitrogenN8OxygenO
What is a physical element?
Physical elements are typically those elements that are exposed in the Physical layer, or in other words, the elements that appear in the right-hand pane of the Administration Tool.
What are the 7 main elements?
The seven element theory included all elements on earth: plants, warm energy, soil, mineral, water, cold energy, and air. This theory introduced each element’s material, property, characteristics, function, relationship to each other, and relationship with life and organs.
How many physical properties are there?
There are six major physical properties. In order for us to measure or observe them, we do not need to change the composition of the substance. The six physical properties are color, density, volume, mass, boiling point, and melting point.
What are properties of liquids?
Liquids have the following characteristics: no definite shape (takes the shape of its container) has definite volume. particles are free to move over each other, but are still attracted to each other.
What are physical properties of metals?
- Metals can be hammered into thin sheets. …
- Metals are ductile. …
- Metals are a good conductor of heat and electricity.
- Metals are lustrous which means they have a shiny appearance.
- Metals have high tensile strength. …
- Metals are sonorous. …
- Metals are hard.
What is physical and chemical?
In a physical change the appearance or form of the matter changes but the kind of matter in the substance does not. However in a chemical change, the kind of matter changes and at least one new substance with new properties is formed. The distinction between physical and chemical change is not clear cut.
What is the difference between chemical and physical properties?
A physical property is a characteristic of a substance that can be observed or measured without changing the identity of the substance. Physical properties include color, density, hardness, and melting and boiling points. A chemical property describes the ability of a substance to undergo a specific chemical change.
What are physical and chemical changes class 9?
1)No new substance is formed in the physical change. A new substance is formed in a chemical change. 2)Physical change is temporary change. A chemical change is a permanent change. … A lot of heat or light energy is absorbed or given out in a chemical change.
What are the chemical properties of metals?
- The density of metals is usually high.
- Metals are malleable and ductile.
- Metals form an alloy with other metals or non – metals.
- Some metals react with air and corrode. …
- Metals are good conductors of heat and electricity. …
- Generally, metals are in a solid state at room temperature.
Is smell a chemical property?
Characteristics such as melting point, boiling point, density, solubility, color, odor, etc. are physical properties. Properties that describe how a substance changes identity to produce a new substance are chemical properties. … Physical and chemical properties can be used to classify a substance as ionic or molecular.
What is a chemical property of an ice cube?
Because they share a common composition with their liquid state, ice molecules also consist of the same 2 to 1 ratio of hydrogen and oxygen atoms, the H2O molecule. The shape of this molecule, the oxygen atom at the center with the two hydrogen atoms separated by an angle of 104.52°, dictates the structure of ice.



