Is the removal of an alcohol oxidation or reduction
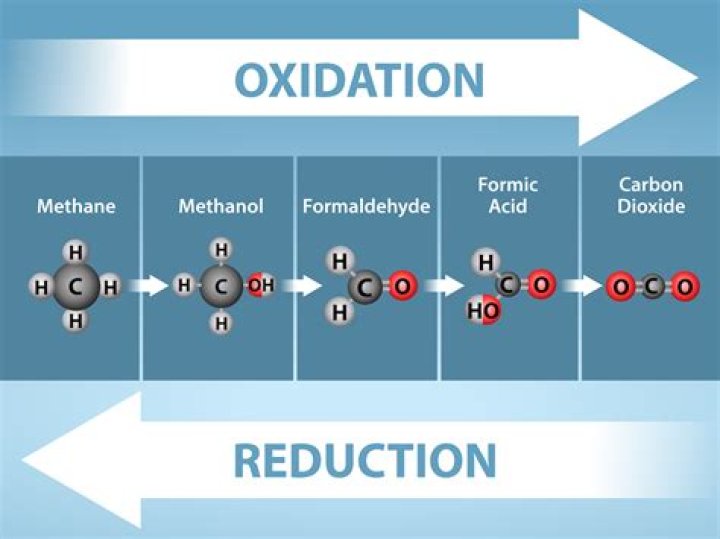
Alcohols may be oxidized to give aldehydes, ketones, and carboxylic acids. The oxidation of organic compounds generally increases the number of bonds from carbon to oxygen, and it may decrease the number of bonds to hydrogen.
Is alcohol reduced or oxidized?
Alcohols may be oxidized to give aldehydes, ketones, and carboxylic acids. The oxidation of organic compounds generally increases the number of bonds from carbon to oxygen, and it may decrease the number of bonds to hydrogen.
What does oxidation remove?
Oxidation is usually used to describe a process in which electrons are removed from a molecule or atom. Here’s an example: … Oxidation means the addition of oxygen to a molecule or the removal of hydrogen from a molecule. Reduction means the addition of hydrogen to a molecule or the removal of oxygen from a molecule.
Can an alcohol be oxidized?
The oxidation of alcohols is an important reaction in organic chemistry. Primary alcohols can be oxidized to form aldehydes and carboxylic acids; secondary alcohols can be oxidized to give ketones. Tertiary alcohols, in contrast, cannot be oxidized without breaking the molecule’s C–C bonds.How do you tell if a reaction is oxidation reduction or neither?
Oxidation numbers represent the potential charge of an atom in its ionic state. If an atom’s oxidation number decreases in a reaction, it is reduced. If an atom’s oxidation number increases, it is oxidized.
What is oxidation of ethanol?
When ethanol is oxidized, it gains an oxygen atom and two additional carbon-oxygen bonds. When ethanol is oxidized, the common oxidizing agent employed is chromic acid, which is an inorganic reagent that is particularly good at oxidizing alcohols and other types of functional groups.
Do alcohols reduce?
Because the most electrophilic site of an alcohol is the hydroxyl proton and because OH- is a poor leaving group, alcohols do not undergo substitution reactions with nucleophiles. The net result of the process is the reduction of alcohols to alkanes. …
Where does oxidation of alcohol take place?
2.8 Conclusions. Alcohol oxidation occurs mainly in the liver, catalyzed by cytosolic ADH and mitochondrial ALDH2 enzymes.What is oxidation in liquor?
Oxidation is one of the main wine faults that sommeliers will see if you recognize as they pour you that little taste after you order a bottle at a restaurant. Too much air makes white wine taste rather nutty and acidic, while turning red into something bitter and tart.
Are alcohols acidic or basic?By the Arrhenius definition of an acid and base, alcohol is neither acidic nor basic when dissolved in water, as it neither produces H+ nor OH- in solution. They are generally weak acids. Alcohols are very weak Brønsted acids with pKa values generally in the range of 15 – 20.
Article first time published onIs ethanol a reducing agent?
Ethanol was observed to act as a reducing agent in the presence of microwave.
Why is reduction called reduction?
Reduction is known as reduction because it refers to addition of electrons, which leads to a reduction in oxidation number of the atom. lol! its called reduction because the opposite of reduction is called oxidation in which oxygen molecule is added in the compound. and when the reducing of that oxygen takes place.
What is oxidation VS reduction?
Oxidation is the gain of oxygen. Reduction is the loss of oxygen.
Is NAD+ oxidized or reduced?
The cofactor is, therefore, found in two forms in cells: NAD+ is an oxidizing agent – it accepts electrons from other molecules and becomes reduced. This reaction, also with H+, forms NADH, which can then be used as a reducing agent to donate electrons. These electron transfer reactions are the main function of NAD.
Which of the following is a reduction reaction?
The correct answer is option 3. Reduction means losing oxygen in a chemical reaction. … 2 H g O ( s ) → h e a t 2 H g ( l ) + O 2 ( g ) , where the oxidation number of Hg is decreasing from +2 in LHS to 0 in RHS.
How do you identify oxidizing and reducing agents?
Break the reaction down into a net ionic equation and then into half-reactions. The substance that loses electrons is being oxidized and is the reducing agent. The substance that gains electrons is being reduced and is the oxidizing agent.
Why is oxidation of alcohols important?
Oxidation of Alcohols to Aldehydes or Ketones: Biological Importance. The oxidation of alcohol groups to carbonyl groups represents an important step in the degradation of fats during the human metabolism (e.g. L- malate to oxaloacetate). Such oxidations are also part of the citric acid cycle.
Can alcohol be reduced by LiAlH4?
LiAlH4 is a strong, unselective reducing agent for polar double bonds, most easily thought of as a source of H-. It will reduce aldehydes, ketones, esters, carboxylic acid chlorides, carboxylic acids and even carboxylate salts to alcohols.
Is a ketone to an alcohol oxidation or reduction?
Because hydride can be thought of as a proton plus two electrons, we can think of conversion of a ketone or an aldehyde to an alcohol as a two-electron reduction. An aldehyde plus two electrons and two protons becomes an alcohol. Aldehydes, ketones and alcohols are very common features in biological molecules.
Is alcohol a chemical change?
Rubbing Alcohol Evaporating This is a physical change because the composition or make up of the substance is not changing. The alcohol is only changing states from liquid to gas. 5.
Why is ethanol oxidized?
Oxidation of alcohols is oxidation in terms of hydrogen transfer. The alcohol is oxidised by loss of hydrogen. Oxidation and reduction in terms of hydrogen transfer is common in hydrocarbon chemistry. Ethanol is oxidised by sodium dichromate (Na2Cr2O7) acidified in dilute sulphuric acid to form the aldehyde ethanal.
Is alcohol a catalyst?
The bicyclic imidazolyl alcohol 1 is a much more efficient catalyst than imidazole (2), that had been previously used in stoichiometric amounts to promote the MBH reactions of cyclic enones in the presence of water.
Can you reduce an alcohol chemistry?
Reduction of Alcohols Normally an alcohol cannot be directly reduced to an alkane in one step. The –OH group is a poor leaving group so hydride displacement is not a good option – however the hydroxyl group is easily converted into other groups that are superior leaving groups, and allow reactions to proceed.
Is ethanol a primary alcohol?
It can also be defined as a molecule containing a “–CH2OH” group. In contrast, a secondary alcohol has a formula “–CHROH” and a tertiary alcohol has a formula “–CR2OH”, where “R” indicates a carbon-containing group. Examples of primary alcohols include ethanol and 1-butanol.
What do you mean by oxidation?
Oxidation is a process in which a chemical substance changes because of the addition of oxygen. The reaction between magnesium metal and oxygen involves the oxidation of magnesium. … Oxidation is a process in which a chemical substance changes because of the addition of oxygen.
When a primary alcohol is completely oxidized The product is?
Primary alcohols are oxidized to form aldehydes and carboxylic acids.
Which of the following alcohol is not oxidized by PCC?
Option A is nothing but phenol, which cannot be oxidized by using PCC. Option A is the correct one. Note: As PCC is a weak oxidizing agent, it cannot oxidize the primary alcohols directly to carboxylic acids but oxidizes primary alcohols to aldehydes only.
Is alcohol a pH?
The pH of alcoholic beverages ranged from 2.49 (Miks Ice Tea – Green Fruits) to 7.64 (Smirnoff). The highest values of acid titration (4.68) and buffer capacity (19.97) were observed in Smirnoff Ice.
Is alcohol a weak acid?
Like water, alcohols are weak acids and bases. This is attributed to the polarization of the O–H bond making the hydrogen partially positive. Moreover, the electron pairs on the oxygen atom of alcohol make it both basic and nucleophilic.
Why are primary alcohols more acidic?
For the simplest case of alkyl alcohols, primary alcohols are more acidic than secondary alcohols which are more acidic than tertiary alcohols. This is because the strength of the alcohol as an acid is dependent on the corresponding strength of its conjugate base, the alkoxide ion.
What is removal of hydrogen?
The removal of hydrogen from a substance is called oxidation.



