Is nitrile a carboxylic acid derivative
Nitriles of Carboxylic Acids Nitriles contain the cyano group, and although they lack the carbonyl group that the other carboxylic acid derivatives have, they are still classified as carboxylic acid derivatives since they are hydrolyzed to carboxylic acids, and also can be produced by dehydration of primary amides.
Is nitrile a carboxylic acid?
Nitriles, RC≡N, are organic cyanides. They are named after the corresponding carboxylic acids by changing -ic acid to -onitrile, or -nitrile, whichever preserves a single letter o. Thus, CH3CN is acetonitrile (from acetic acid), whereas C6H5CN is benzonitrile (from benzoic acid).
How do carboxylic acids form from nitrile?
Acid hydrolysis The nitrile is heated under reflux with a dilute acid such as dilute hydrochloric acid. A carboxylic acid is formed. For example, starting from ethanenitrile you would get ethanoic acid. The ethanoic acid could be distilled off the mixture.
What are all of the carboxylic acid derivatives?
The functional groups at the heart of this chapter are called carboxylic acid derivatives: they include carboxylic acids themselves, carboxylates (deprotonated carboxylic acids), amides, esters, thioesters, and acyl phosphates. Cyclic esters and amides are referred to as lactones and lactams, respectively.What are carboxylic acid derivatives define?
Carboxylic acid derivatives are compounds with the acyl group, RCO-, bonded to an electronegative atom or substituent, -Y, that can act as a leaving group in substitution reactions (nucleophilic acyl substitution). 2. Carboxylic acid derivatives are compounds that yield a carboxylic acid upon rxn with water.
How do you convert an ester to a carboxylic acid?
The conversion of ester into acid is by achieved by the hydrolysis of ester in presence of alkali in alcoholic soln. under reflux then neutralization by acid salt to get carboxylic acid.
Is ester a carboxylic acid derivative?
The reaction of an acyl chloride with an alcohol also gives an ester, but this conversion cannot be reversed by adding HCl to the reaction mixture. Esters are one of the most common carboxylic derivatives.
How do you name carboxylic acid derivatives?
The nomenclature of acid halides starts with the name of the corresponding carboxylic acid. If the corresponding carboxylic acid has an –oic acid or –ic acid ending it is removed and replaced with the ending -oyl followed by the first syllable of the name of the halogen along with an –ide ending.Which is not the derivative of carboxylic acid?
Which of the following is not a derivative of a carboxylic acid? Explanation: Aldehyde is not a derivative of carboxylic acid. Esters can be derived from carboxylic acids by reacting them with to form an acid chloride.
How are carboxylic acid derivatives obtained?Carboxylic acids have a hydroxyl group bonded to an acyl group, and their functional derivatives are prepared by replacement of the hydroxyl group with substituents, such as halo, alkoxyl, amino and acyloxy. The carboxylic acid derivatives can all be hydrolyzed to carboxylic acids.
Article first time published onWhat kind of intermolecular bonding occurs between carboxylic acids?
Carboxylic acids, similar to alcohols, can form hydrogen bonds with each other as well as van der Waals dispersion forces and dipole-dipole interactions.
Which reagent would convert a nitrile to a carboxylic acid?
Nitriles can be converted to carboxylic acid with heating in sulfuric acid. During the reaction an amide intermediate is formed.
How is acetic acid prepared from nitrile?
Answer: You can convert acetonitrile (ethanenitrile) to acetic acid by acid hydrolysis – reflux the nitrile with dilute hydrochloric acid, and you will form the acetic acid which can then be distilled off.
What are the four acid derivatives?
Although there are many types of carboxylic acid derivatives known we will be focusing on just four: Acid halides, Acid anhydrides, Esters, and Amides.
What is the other name of ester interchange reaction?
What is the other name of ester-interchange reaction? Explanation: In the alcoholysis or ester-interchange reaction, an alcohol reacts with an ester to give a new ester. 2.
Why is an ester defined as a carboxylic acid derivative?
Esters and amides are considered to be derivatives of carboxylic acids because the OH in the carboxyl group is replaced with another group.
Are esters more reactive than carboxylic acids?
Under the reaction condition s the carboxylic acid will deprotonate to give the carboxylate which is a very poor electrophile (after all, it has a negative charge !) so the ester is more reactive than the acid.
Which of the following acid is a derivative of dicarboxylic acid?
The most widely used dicarboxylic acid in the industry is adipic acid, which is a precursor used in the production of nylon. Other examples of dicarboxylic acids include aspartic acid and glutamic acid, two amino acids in the human body. The name can be abbreviated to diacid.
What type of reaction is ester to carboxylic acid?
Conversion of Esters to Carboxylic Acids: Saponification The reaction is commonly called a saponification from the Latin sapo which means soap.
What is the other product formed when an ester is made by reacting a carboxylic acid and an alcohol?
Esters and water are formed when alcohols react with carboxylic acids. This reaction is called esterification, which is a reversible reaction. … Since esterification is a reversible reaction, esters can undergo hydrolysis to form corresponding alcohol and organic acid.
What are acid derivatives?
Carboxylic acid derivative: A carbonyl-containing functional group other than an aldehyde or ketone, or a molecule containing such a functional group. Called a carboxylic acid derivative because these functional groups can, in principle, be produced (derived) from the corresponding carboxylic acid.
Which of the following is not a carboxylic acid *?
Picric acid is an organic compound with the formula (O2N)3C6H2OH. Its IUPAC name is 2,4,6-trinitrophenol (TNP). It is one of the most acidic phenols (alcohols). Like other highly nitrated organic compounds, picric acid is highly explosive.
What is the prefix of carboxylic acid?
Functional groupPrefixSuffixcarboxylic acidsnone-oic acidaldehydesnone-alketonesnone-onealcholshydroxy–ol
How do you identify the functional group of a carboxylic acid?
Prepare a saturated solution of sodium bicarbonate by dissolving sodium bicarbonate in 1ml of water. Add the given organic compound on the saturated solution of sodium bicarbonate solution. Shake the solution well. If there is an evolution of brisk effervescence then it indicates the presence of carboxylic acid.
Do esters have stronger intermolecular forces than carboxylic acids?
Ester molecules are polar but have no hydrogen atom attached directly to an oxygen atom. They are therefore incapable of engaging in intermolecular hydrogen bonding with one another and thus have considerably lower boiling points than their isomeric carboxylic acids counterparts.
Why do carboxylic acids have stronger intermolecular forces?
Carboxylic acids show a high degree of association through hydrogen bonding. We have encountered such bonding previously with alcohols; however, acids form stronger hydrogen bonds than alcohols because their O−H bonds are more strongly polarized as −δ⊖O−δ⊕H.
Is ester a base or acid?
Esters are neutral compounds, unlike the acids from which they are formed. In typical reactions, the alkoxy (OR′) group of an ester is replaced by another group. One such reaction is hydrolysis, literally “splitting with water.” The hydrolysis of esters is catalyzed by either an acid or a base.
Do amines react with carboxylic acids?
The direct reaction of a carboxylic acid with an amine would be expected to be difficult because the basic amine would deprotonate the carboxylic acid to form a highly unreactive carboxylate.
What is chiral carboxylic acid?
Chiral carboxylic acids exist widely in nature in the form of amino acids and compounds such as tartaric acid, malic acid, cholic acid, biotin, etc. … Remarkably, enzymes can easily realize extremely challenging acid–base chemistry in the absence of highly acidic functionality.
Is anhydride a carboxylic acid derivative?
Compounds in which the −OH group of the carboxylic acid is replaced by other functional groups are called carboxylic acid derivatives, the most important of which are acyl halides, acid anhydrides, esters, and amides.
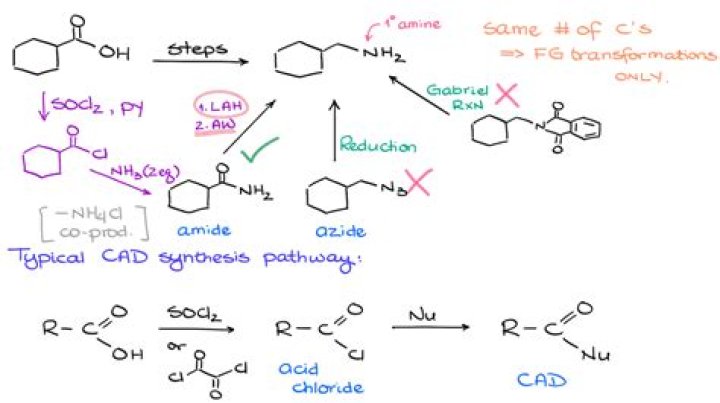
How do you convert nitrile to amine?
Nitriles can be converted to 1° amines by reaction with LiAlH4. During this reaction the hydride nucleophile attacks the electrophilic carbon in the nitrile to form an imine anion. Once stabilized by a Lewis acid-base complexation the imine salt can accept a second hydride to form a dianion.