Is a quadruple bond possible
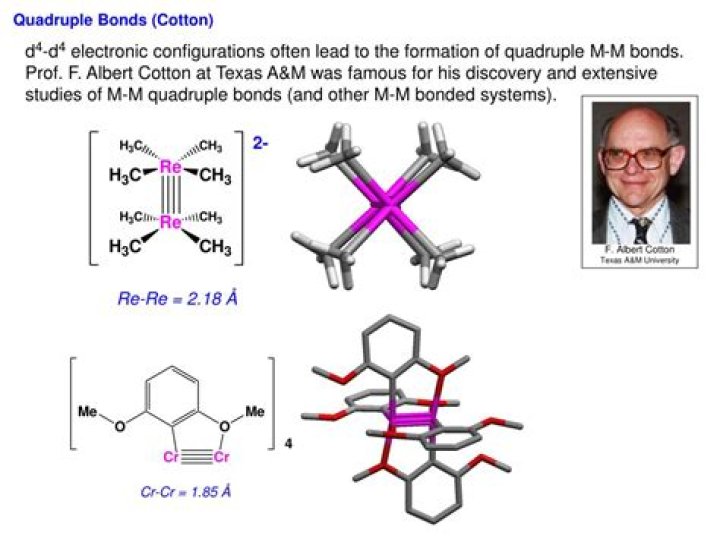
Quadruple (and quintuple) bond is only possible using d orbitals. With them, it can make one σ bond (involving orbitals) , two π bonds ( + orbitals and + orbitals) and two δ (delta) bonds ( + orbitals and + orbitals).
Why is a quadruple bond not possible?
If we go on to the valence-bond model, in which bonds result from the overlap of atomic orbitals, we see a better explanation: carbon cannot form a quadruple bond because it doesn’t have enough atomic orbitals pointing in the right directions.
Why is a quadruple bond not possible between two carbon atoms?
Basically, the orbitals that allow for single, double, and triple bonds between carbons to not have to bend very much, but the last orbital which would allow for a quadruple bond would have to been too far, so it makes it impossible for carbon to form a quadruple bond with itself.
Is it possible to have a quadruple bond in chemistry?
A quadruple bond is a type of chemical bond between two atoms involving eight electrons. … Stable quadruple bonds are most common among the transition metals in the middle of the d-block, such as rhenium, tungsten, technetium, molybdenum and chromium.Is a quadruple covalent bond the strongest?
Double and triple covalent bonds are stronger than single covalent bonds and they are characterized by the sharing of four or six electrons between atoms, respectively.
Why C2 is not possible?
c2 molecule exist in space as gas, but under normal environment it cannot exist as 4 electron bonding with another 4 electron (quadruple bond) is not stable due to large repulsion between electron (same charge repel) and is very unstable.
Are quintuple bonds possible?
A quintuple bond in chemistry is an unusual type of chemical bond, first reported in 2005 for a dichromium compound. Single bonds, double bonds, and triple bonds are commonplace in chemistry.
Does B2 have a triple bond?
So the bond order of B2 is equal to 1, which you can get by drawing the molecular orbital diagram and performing the equation Bond Order = . 5 * (# of bonding electrons – # of antibonding electrons). However, when you draw the Lewis structure of B2, you get a triple bond.What kind of bonds are present in Cao?
Ca is a metal while O is a nonmetal. This makes them an ionic compound. Recall that covalent molecules are made up of nonmetals only.
Is diatomic carbon possible?Carbon is not a diatomic element. Diatomic elements are those where two atoms of the element can join to form a single molecule.
Article first time published onWhy does diatomic carbon not exist?
Carbon is not diatomic because it’s not able to form stable* quadruple bonds. In order to form a quadruple bond according to low-level, simple-minded MO theory, carbon would need to have occupied d-orbitals that could overlap to form a bond.
Can carbon form 4 bonds with another carbon?
Carbon can not form 4 bonds with another carbon because of its orbitals which are some time hybridized.
What are the only elements that exist in nature as uncombined atoms?
In nature, only the noble gas elements, such as helium and neon, exist as uncombined atoms. They are monatomic; that is, they consist of single atoms.
Are triple bonds the longest?
The bond length depends on the strength of the bond. The stronger the bond is, the shorter it will be. The triple bonds are the strongest and hence the shortest. … Finally, single bonds are the longest among the three.
Which bond is the strongest bond?
In chemistry, covalent bond is the strongest bond. In such bonding, each of two atoms shares electrons that binds them together. For example, water molecules are bonded together where both hydrogen atoms and oxygen atoms share electrons to form a covalent bond.
What type of chemical bond is the weakest?
The weakest of the intramolecular bonds or chemical bonds is the ionic bond then polar covalent bond and the strongest is the non-polar covalent bond.
Who discovered the quintuple bond?
Albert Cotton and his coworkers at Texas A&M University surprised chemists in 1964 with evidence that the [Re2Cl8]2­ ion contained the first known multiple bond between two metal atoms.
How many bonds can chromium form?
(You can count six bonds and one interaction per chromium atom.) So it needs to use 6 orbitals to bond in this compound. Valence orbitals are for holding valence electrons, and valence electrons are used for bonding.
What type of bond is Cu Zn alloy?
These two elements are both metals and they will form Metallic bonds when reacted to each other.
Does Dicarbon exist?
Carbon, the sixth most abundant element in the universe, has been known since ancient times. … Three naturally occurring allotropes of carbon are known to exist: amorphous, graphite and diamond. Amorphous carbon is formed when a material containing carbon is burned without enough oxygen for it to burn completely.
Does Ne2 exist?
Ne2 molecule does not exist .
Does c4 exist?
C-4 or Composition C-4 is a common variety of the plastic explosive family known as Composition C, which uses RDX as its explosive agent. C-4 is composed of explosives, plastic binder, plasticizer to make it malleable, and usually a marker or odorizing taggant chemical.
What type of bond is CU?
Copper wire consists of Cu nuclei (that are positively charged due to the positively charged protons) that are surrounded by a “sea” of delocalized electrons (that are negatively charged that move freely throughout the material which is known as metallic bonding.
How many bonding pairs does Cao have?
Each oxygen contributes 2 electrons – 1 for each bond. That means there are a total of 8 electrons around the carbon, in 4 pairs. Because there are 4 bonds, these are all bondpairs.
What is meant by Cao?
From Wikipedia, the free encyclopedia. Chief administrative officer. Synonyms. CAO. A chief administrative officer (CAO) is a top-tier executive who supervises the daily operations of an organization and is ultimately responsible for its performance.
Can H2+ exist?
The hydrogen atom have only one electron, how could it turn to H2+? H2+ does not exist, so the question is unmeaning.
Can pi bonds exist without sigma bond?
A pi bond can exist between two atoms that do not have a net sigma-bonding effect between them. In certain metal complexes, pi interactions between a metal atom and alkyne and alkene pi antibonding orbitals form pi-bonds.
Can B2 exist?
Magnetic property: Since bond order is zero, Be2 molecule does not exist. It is diamagnetic due to the absence of any unpaired electron. B2 molecule is formed by the overlap of atomic orbitals of both boron atoms. … The two boron atom is B2 molecules are linked by one covalent bond.
Does He2 exist?
He2 does not exist because it has filled octets and its valency is zero. Because it has no combining capacity, it exists as a monoatomic molecule like He. The electrical configuration of the helium atom is 1s2.
Does c2 exist according to MOT?
Hint: Recall the molecular orbital theory (MOT) and write the electronic configuration of ${C_2}$ molecule according to MOT. You will find that the ${C_2}$ molecule has two sets of paired orbitals in the degenerate pi-bonding orbitals and bond order comes out to be 2.
Which molecule Cannot exist as a diatomic?
Hydrogen exists as a diatomic molecule in its elemental form; helium does not exist as a diatomic molecule.