How many protons are in the nucleus of radon
In either case, it simply designates the element radon, which always has 86 protons, and that the particular isotope of radon in question is the one with 136 neutrons.
How many proton are in a nucleus?
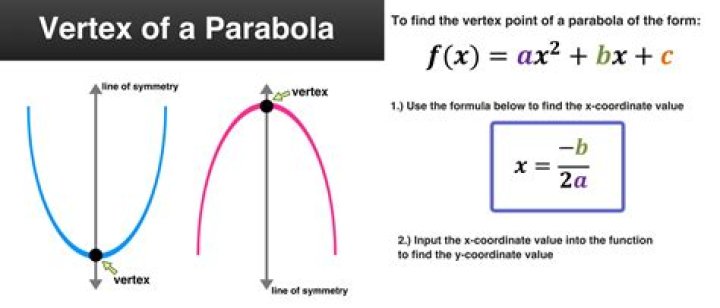
The number of protons in the nucleus of the atom is equal to the atomic number (Z). The number of electrons in a neutral atom is equal to the number of protons. The mass number of the atom (M) is equal to the sum of the number of protons and neutrons in the nucleus.
How many protons neutrons and electrons are in the neutral atom radon 220 radon Rn?
Rn has 86 electrons outside the nucleus, it has 86 protons and (222-86 =136) neutrons inside the nucleus. Rn has 86 electrons outside the nucleus and has 86 protons and (220-86 = 134) neutrons inside the nucleus.
How many protons are in the nucleus of radium?
A radium nucleus is a massive nucleus of 226 nucleons, including 88 protons and 138 neutrons. It decays by emitting an alpha particle composed of two protons and two neutrons.What element has 4 protons 4 neutrons and 2 electrons?
NameBerylliumSymbolBeProtons4Neutrons5Electrons4
How many protons does radon 226 have?
The atomic number, 88, is the number of protons. The mass number, 226, is the total number of protons and neutrons. particle (2 protons and 2 neutrons).
What do protons do in the nucleus?
Protons define the entire charge of a nucleus, and hence its chemical identity. Neutrons are electrically neutral, but contribute to the mass of a nucleus to nearly the same extent as the protons. Neutrons can explain the phenomenon of isotopes (same atomic number with different atomic mass).
How many protons are in the nucleus of radon 222?
For example, all radon isotopes have 86 protons (Z=86), but radon-222 has 136 neutrons (86 + 136 = 222), whereas radon- 220 has only 134 neutrons (86 + 134 = 220). The chemical symbol for radon is Rn, and the mass number is usually placed either after the symbol (Rn-222) or to the left and above it (222Rn).How many electrons and protons does radium have?
Atomic Number88Number of Electrons (with no charge)88Number of Protons88Mass Number226Number of Neutrons138
How many protons and electrons does radon have?The nucleus consists of 86 protons (red) and 136 neutrons (orange). 86 electrons (white) successively occupy available electron shells (rings). Radon is a noble gas in group 18, period 6, and the p-block of the periodic table. It is the only known radioactive gas.
Article first time published onWhat has 14 protons in the nucleus?
For example, silicon has 14 protons and 14 neutrons. Its atomic number is 14 and its atomic mass is 28.
Which element has 16 protons in its nucleus?
On the periodic table of elements, sulfur is represented by the symbol S. It has an atomic number of 16 because it has 16 protons in its nucleus.
Where are protons located?
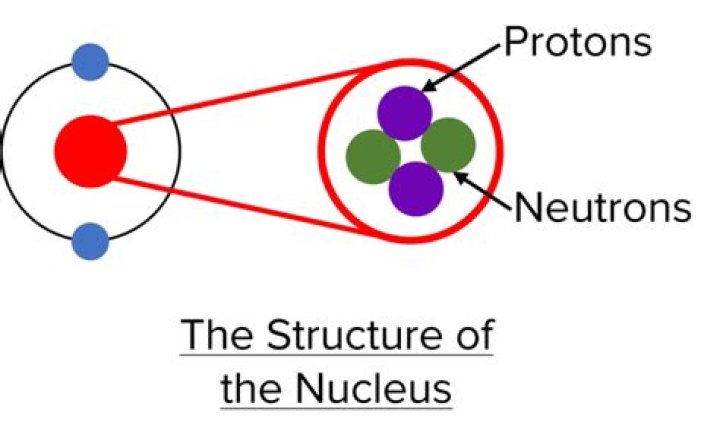
Atomic particles Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom.
What is meant by proton number?
The proton number (Z) of an atom is the number of protons it contains. All the atoms of a particular element have the same proton number (number of protons). … The nucleon number (or mass number) of an atom is the total number of protons and neutrons it contains.
Are protons outside the nucleus?
Protons are among the (most, least) massive subatomic particles, and they are found (inside, outside) the nucleus.
What is electron neutron and proton?
Atoms consist of three basic particles: protons, electrons, and neutrons. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). The outermost regions of the atom are called electron shells and contain the electrons (negatively charged).
How does a proton and neutron bind?
Protons and neutrons are made up of smaller subatomic particles. When protons or neutrons get close enough to each other, they exchange particles (mesons), binding them together. … Although the strong force overcomes electrostatic repulsion, protons do repel each other.
How many protons does radon 220?
Iso- topes have the same number of protons (and therefore they are the same element), but they have different numbers of neutrons. For example, all radon isotopes have 86 protons (Z=86), but radon-222 has 136 neutrons (86 + 136 = 222), whereas radon-220 has only 134 neutrons (86 + 134 = 220).
How many protons does the element Xe have?
Xenon is a clear and colorless, and odorless gas that is quite heavy. Xenon gas is 4.5 times heavier than Earth’s atmosphere (which consists of a mixture of a number of gaseous elements and compounds). This element’s mass comes from its nucleus, which contains 54 protons and a varying (but similar) number of neutrons.
What element has 82 protons in the nucleus?
On the periodic table of elements, lead is represented by the symbol Pb. It has an atomic number of 82 because it has 82 protons in its nucleus.
What is the proton and neutron of radium?
The nucleus consists of 88 protons (red) and 138 neutrons (orange). 88 electrons (white) successively occupy available electron shells (rings). Radium is a radioactive alkaline earth metal in group 2, period 7, and the s-block of the periodic table. Formed in uranium ores, the Curies discovered it in 1898.
How many electrons does radon have in its outer shell?
Radon atoms possess a particularly stable electronic configuration of eight electrons in the outer shell, which accounts for the characteristic chemical inactivity of the element. Radon, however, is not chemically inert.
Is radon a metal?
Classification:Radon is a noble gas and a nonmetalAtomic weight:(222), no stable isotopesState:gasMelting point:-71 oC, 202 K
How many protons does RN 224 have?
Radiation Toxicology, Ionizing and Nonionizing In Europe, the short-lived radionuclide (half-life 3.6 days) radium-224 was used for more than 40 years in the early 1900s in treating tuberculosis and ankylosing spondylitis.
How do u find neutrons?
To find the number of neutrons, subtract the number of protons from the mass number. number of neutrons=40−19=21.
What is radon-222 Half-Life?
Rn-222 has a short half-life (3.8 days) and decays into a series of solid particulate products, known as radon progeny or radon daughters, all of which have even shorter half-lives (∼30 min or less).
What does the 222 in radon-222 represent?
Element NameRadonElement SymbolRnAtomic Number86
How many neutrons are in CA?
Johnson Z. In calcium-40, there are 20 neutrons.
What has 5 protons and 5 neutrons?
NameProtonsNeutronsHelium22Lithium34Beryllium45Boron56
How do you find the protons neutrons and electrons?
To calculate the numbers of subatomic particles in an atom, use its atomic number and mass number: number of protons = atomic number. number of electrons = atomic number.
How many protons neutrons and electrons does xenon have?
The nucleus consists of 54 protons (red) and 78 neutrons (yellow). 54 electrons (white) bind to the nucleus, filling the outer (fifth) electron shell in what is a very stable configuration. The stability of an element’s outer electrons determines its chemical and physical properties.