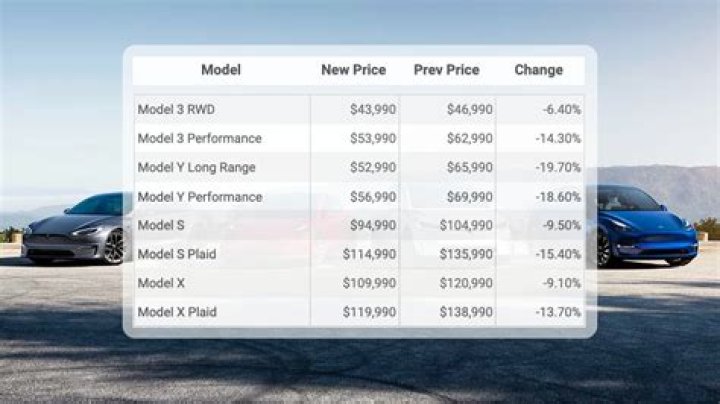
How many electrons are in the 4th energy level in the Ga atom

The fourth energy level has 18 electrons. The fourth energy level of the periodic table includes the 4s 3d and 4p orbitals. The 4p orbital holds 6 electrons. There is a 4d orbital with 10 electrons which coincides with the 5th energy level of the periodic table.
How many electrons does 4 energy level?
Regardless of its shape, each orbital can hold a maximum of two electrons. Energy level I has just one orbital, so two electrons will fill this energy level. Energy level IV has 16 orbitals, so it takes 32 maximum electrons to fill this energy level.
How many electrons are in 4th shell?
n=ShellMaximum Number of Electrons11st Shell222nd Shell833rd Shell1844th Shell32
How many p electrons are there in an atom of gallium Ga?
It has 31 electrons. We form cations by removing electrons first from the outermost p orbitals, followed by the s orbital and finally the d orbitals (if any more electrons need to be removed).Which element has electrons in 4 energy levels?
Atomic NumberElementEnergy Levels or “shells”4Beryllium (Be)25Boron (B)36Carbon (C)47Nitrogen (N)5
What has 3 electrons in n 4 energy level?
Orbitals and Electron Capacity of the First Four Principle Energy LevelsPrinciple energy level (n)Type of sublevelNumber of orbitals per type3p3d54s1
What is the 4th energy level?
The fourth energy level has 18 electrons. The fourth energy level of the periodic table includes the 4s 3d and 4p orbitals. The 4p orbital holds 6 electrons. There is a 4d orbital with 10 electrons which coincides with the 5th energy level of the periodic table.
What ion does Ga form?
PubChem CID105145StructureFind Similar StructuresMolecular FormulaGa+3SynonymsGallium cation gallium(3+) Gallium (III) ion Gallium, ion(3+) UNII-F7K5MP217W More…Molecular Weight69.723How many electrons are in the N 4 level of a ground state gallium GA z 31 atom?
Gallium atoms have 31 electrons and the shell structure is 2.8. 18.3. The ground state electron configuration of ground state gaseous neutral gallium is [Ar].
How many electrons are in 4th shell GCSE?Energy shellMaximum number of electronsSecond8Third8
Article first time published onHow many valence electrons are in the 4th shell?
In my textbook, it says that the maximum number of electrons that can fit in any given shell is given by 2n². This would mean 2 electrons could fit in the first shell, 8 could fit in the second shell, 18 in the third shell, and 32 in the fourth shell.
What is number 4 on the periodic table?
Beryllium is the element that is atomic number 4 on the periodic table.
What element has 4 energy levels and 5 valence electrons?
Explanation: The elements of group 15 (column) VA of the periodic table all have electron configurations of s2p3 , giving them five valence electrons. These elements include Nitrogen (N), Phosphorus (P), Arsenic (As), Antimony (Sb) and Bismuth (Bi). [Ar]4s23d104p3 .
What element has 4 electrons in Shell 2?
ZElementNo. of electrons/shell3Lithium2, 14Beryllium2, 25Boron2, 36Carbon2, 4
How many orbitals are possible in the 4th energy level?
Total number of orbitals = total values of ml For n=4, ∴1+3+5+7=16 orbitals present in 4th energy level.
What are the 4 electron orbitals?
Current theory suggests that electrons are housed in orbitals. An orbital is a region of space where there is a high probability of finding an electron. There are four basic types of orbitals: s, p, d, and f. An s orbital has a spherical shape and can hold two electrons.
What is the maximum number of electrons in the principal energy level 4?
Thus, the fourth level can hold up to 32 electrons: 2 in the s orbital, 6 in the three p orbitals, 10 in the five d orbitals, and 14 in the seven f orbitals. The sublevels of the first four principal energy levels and the maximum number of electrons that the sublevels can contain are summarized in Table 5.1.
How many sublevels are there with N 4 in an atom?
We have 4− subshells in this case; s,p,d,f ↔ 0,1,2,3 for the value of l . =16−− orbitals in the n=4− energy level.
How do you find number of electrons?
To calculate the numbers of subatomic particles in an atom, use its atomic number and mass number: number of protons = atomic number. number of electrons = atomic number.
How many electrons are there in CA?
Ca has 20 protons, so neutral it would have 20 electrons, but according to the charge, 2 electrons have been lost.
How much electrons are in GA?
Gallium Atomic and Orbital Properties Gallium atoms have 31 electrons and the electronic shell structure is [2, 8, 18, 3] with Atomic Term Symbol (Quantum Numbers) 2P1/2.
How many 3d electrons are in GA?
Okay, so a neutral gallium will have 31 electrons now, gallium three. Plus, that means it has lost three electrons, right? Because it’s plus, So instead of having 31 electrons is going to have 31 minus three, which is 28 electrons.
How many valence electrons are in GA?
Gallium has the following electron configuration. The 4s and 4p electrons can be lost in a chemical reaction, but not the electrons in the filled 3d subshell. Gallium therefore has three valence electrons.
How many neutrons does SI 4 -) have?
Silicon has 14 protons, 14 neutrons, and 14 electrons.
How many ions Does Ga have?
In most of its compounds, gallium has an oxidation state of +3 and, in a few, +1 (for example, the oxide, Ga2O). There is no evidence for authentic compounds of gallium in its +2 state. The “dihalides,” for example, contain Ga+ and Ga3+ in a one-to-one ratio.
How many protons are in Ga 3+?
We’re taking a look at an ion to determine the number of protons and electrons that it has here that is gallium galleons in Group three. So that’s why it’s three. If we take a look at the period, we’ll see that it has a number of 31 means It has 31 proton now here.
Is Ga an atom or ion?
Density (near r.t. ) when liquid (at m.p. ) Gallium is a chemical element with the symbol Ga and atomic number 31. Discovered by French chemist Paul-Émile Lecoq de Boisbaudran in 1875, Gallium is in group 13 of the periodic table, and has similarities to the other metals of the group (aluminium, indium, and thallium).
Where is Group 4 on the periodic table?
Group 4 is the second group of transition metals in the periodic table. It contains the four elements titanium (Ti), zirconium (Zr), hafnium (Hf), and rutherfordium (Rf). The group is also called the titanium group or titanium family after its lightest member.
What element has 4 protons 4 neutrons and 2 electrons?
NameBerylliumSymbolBeProtons4Neutrons5Electrons4
What element has 4 protons 4 electrons and 5 neutrons?
4 protons, 5 neutrons, and 4 electrons are there in an atom of beryllium.
What element has 4 energy levels and 4 valence electrons?
CARBON FAMILY: All have 4 VALENCE ELECTRONS (electrons in the outer energy level) Carbon is a NONMETAL. Silicon and Germanium are METALLOIDS.