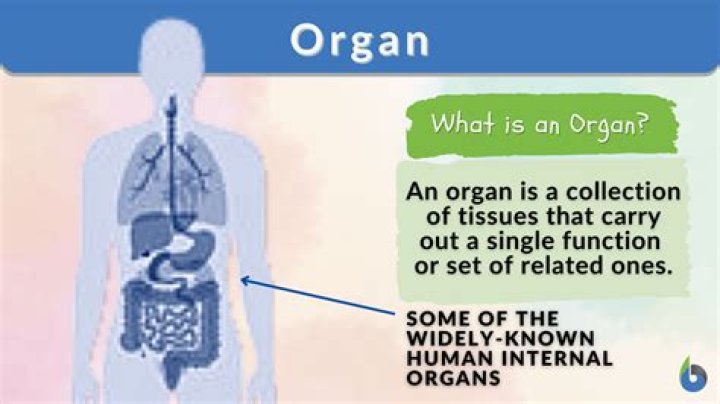
How does the furnace get the energy needed to heat the water

A hot-water heating system burns fuel in a boiler to produce thermal energy. The thermal energy is used to heat water, which is pumped through a system of pipes and radiators. A warm-air heating system burns fuel in a furnace to produce thermal energy.
What is the energy required to heat water?
The specific heat capacity of water is 4,200 Joules per kilogram per degree Celsius (J/kg°C). This means that it takes 4,200 J to raise the temperature of 1 kg of water by 1°C.
How does energy get converted to heat?
An electric heater is an electrical appliance that converts electrical energy into heat. The heating element inside every electric heater is simply an electrical resistor, and works on the principle of Joule heating: an electric current through a resistor converts electrical energy into heat energy.
How does a furnace transfer energy?
Furnaces employing combustion produce a hot gas which transfers heat to the material by radiation and convection. Solids are heated by direct contact, but fluids are usually heated indirectly, being carried inside pipes within the furnace.How is the thermal energy of the radiator transferred to the surrounding air?
The radiator transfers thermal energy to the air around it by conduction and radiation. The warm air then circulates throughout the room in convection currents. The hot water cools as it flows through the system and transfers its thermal energy.
How do you calculate the energy required to heat water?
The specific heat capacity of water is 4.18 J/g/°C. We wish to determine the value of Q – the quantity of heat. To do so, we would use the equation Q = m•C•ΔT. The m and the C are known; the ΔT can be determined from the initial and final temperature.
How do you calculate the energy needed to heat water?
Calculate the kilowatt-hours (kWh) required to heat the water using the following formula: Pt = (4.2 × L × T ) ÷ 3600. Pt is the power used to heat the water, in kWh. L is the number of liters of water that is being heated and T is the difference in temperature from what you started with, listed in degrees Celsius.
How does a furnace work?
A forced-air furnace heats your home through a heating cycle that looks like this: Natural gas or propane is ignited in the burner. The flames heat up a metal heat exchanger and exhaust out of the flue. … The furnace’s blower forces the heated air into the ductwork and distributes it throughout the home.Does a furnace heat water?
Furnaces heat air and distribute the heated air through the house using ducts. Boilers heat water, and provide either hot water or steam for heating. Steam is distributed via pipes to steam radiators, and hot water can be distributed via baseboard radiators or radiant floor systems, or can heat air via a coil.
What is furnace function?Furnaces are mostly used as a major component of a central heating system. The name derives from Latin word fornax, which means oven. Furnaces are permanently installed to provide heat to an interior space through intermediary fluid movement, which may be air, steam, or hot water.
Article first time published onHow do we get electricity from water?
“A hydraulic turbine converts the energy of flowing water into mechanical energy. A hydroelectric generator converts this mechanical energy into electricity. The operation of a generator is based on the principles discovered by Faraday.
How is electric energy converted to heat energy?
Electric heating is a process in which electrical energy is converted to heat energy. … The heating element inside every electric heater is an electrical resistor, and works on the principle of Joule heating: an electric current passing through a resistor will convert that electrical energy into heat energy.
How is energy converted through heat?
Energy transformation, also known as energy conversion, is the process of changing energy from one form to another. … For example, to heat a home, the furnace burns fuel, whose chemical potential energy is converted into thermal energy, which is then transferred to the home’s air to raise its temperature.
Do refrigerators have thermal energy?
Overview. A refrigerator is an example of a cooling system. The purpose of any cooling system is to transfer thermal energy in order to keep things cool. A refrigerator transfers thermal energy from the cool air inside the refrigerator to the warm air in the kitchen.
What happens to the steam as it gives up thermal energy?
CONDENSATION When steam is cooled, it releases thermal energy and turns into its liquid state. This process is called condensation. reversed if the liquid is cooled. The change from the liquid state to the solid state is called freezing.
How do you calculate the energy required to heat?
The specific heat capacity is the heat or energy required to change one unit mass of a substance of a constant volume by 1 °C. The formula is Cv = Q / (ΔT ⨉ m) .
How do you calculate water energy?
Using the equation Q=mcΔT we can calculate the amount of energy for heating the water to 100 degrees. c=4187 Joules per kilogram- the specific heat capacity of water.
How do you calculate joules to heat water?
Answer: The specific heat capacity of water is (roughly) 4.184 kilojoules / kg K. (10 kg) × (40 degrees Celsius temperature change) × (4.184 kJ / kg K) = 1673.6 kilojoules.
How do we calculate energy?
In classical mechanics, kinetic energy (KE) is equal to half of an object’s mass (1/2*m) multiplied by the velocity squared. For example, if a an object with a mass of 10 kg (m = 10 kg) is moving at a velocity of 5 meters per second (v = 5 m/s), the kinetic energy is equal to 125 Joules, or (1/2 * 10 kg) * 5 m/s2.
How much energy does it take to heat a gallon of water?
A British Thermal Unit (BTU) is a measurement of heat energy. One BTU is the amount of heat energy required to raise one pound of water by 1ºF. Water weighs 8.33 pounds per gallon so we can calculate that one gallon of water requires 8.33 BTU to raise the temperature 1ºF.
How does hot water furnace work?
The water in the furnace heats to a boil. As it heats up, pressure builds. Once the pressure is at its maximum, the water turns to steam. The pressure also pushes the steam throughout the house.
Does a furnace use water?
Although a furnace doesn’t use water to run, special high efficiency furnace produces condensation during combustion. … It’s this second combustion chamber that allows high-efficiency furnaces can produce more heat energy from their fuel.
How does a water heater heat water?
A gas powered water heater has cold water brought into the tank through a dip tube (1). This water is heated with a gas burner (2). … As this chimney heats up, the surrounding water is heated as well. Heat rises, and water heaters use this to bring warm water through the plumbing of your home.
How does a furnace thermostat work?
Mechanical thermostats regulate heat thanks to the expansion of their internal metallic strips. This strip carries electricity through the connected circuit, switching on the heating. As the strip heats up, one of the metals expands enough to open the circuit and switch off the heating, cooling the room down.
How does a furnace heat a house?
A furnace works by blowing heated air through ducts that deliver the warm air to rooms throughout the house via air registers or grills. … Inside a gas- or oil-fired furnace, the fuel is mixed with air and burned. The flames heat a metal heat exchanger where the heat is transferred to air.
How does a heat exchanger work?
Heat exchangers, metal shells and tubes, work by transferring heat from one place to another. … The hot flue gas heats the metal as the gas makes its way to the exhaust outlet of the furnace. As this is happening, the hot metal heats the air circulating over the exterior of the heat exchanger.
How hot does a furnace get?
A mid-efficiency gas furnace can raise the temperature to 170F, but a high-efficiency model can only raise it to 150F. There are top quality furnaces that can raise it to 200F.
How hot is a furnace?
With a conventional gas furnace, you should be at about 140°F-170°F at the supply plenum. A high efficiency furnace will be lower, around 110°F-140°F. However, the temperature at the outlet of the furnace, is dependent on the temperature of the incoming air.
Does central heating use water?
Central heating should not significantly increase your water usage. Though depending on what type of central heating you’re talking about, it may consume some water.
What is water energy?
Water energy is energy derived from the power of water, most often its motion. Energy sources using water have been around for thousands of years in the form of water clocks and waterwheels. A more recent innovation has been hydroelectricity, or the electricity produced by the flow of water over dams.
What is electricity generated from water called?
Hydroelectricity, or hydroelectric power, is electricity produced from hydropower.