How does temperature affect thermal energy
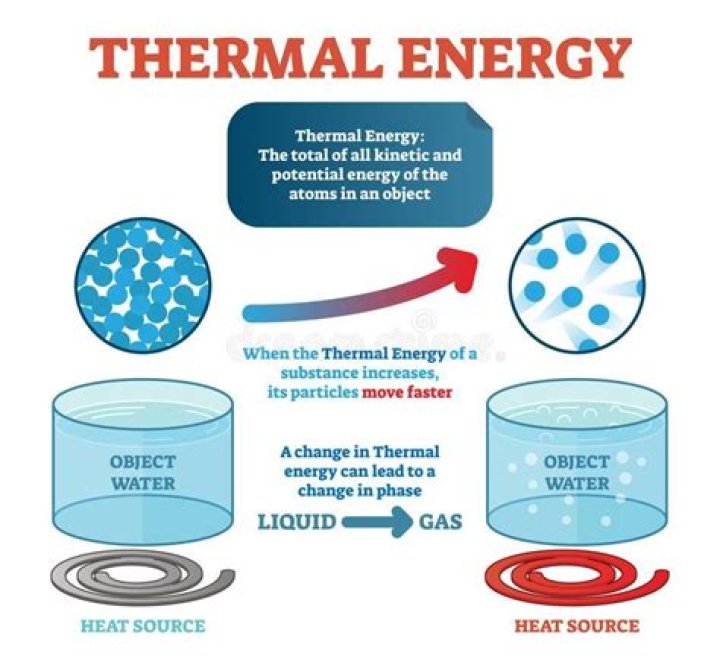
When the temperature of an object increases, the average kinetic energy of its particles increases. When the average kinetic energy of its particles increases, the object’s thermal energy increases. Therefore, the thermal energy of an object increases as its temperature increases.
How does temperature and mass affect thermal energy?
Warmer objects have faster particles and higher temperatures. If two objects have the same mass, the object with the higher temperature has greater thermal energy.
How does decreased temperature affect thermal energy?
If the temperature is increased the particles gain more kinetic energy or vibrate faster. This means that they move faster and take more space. If the temperature is decreased,it is the opposite. Particles move slower, because of less energy.
How does temperature difference affect thermal energy transfer?
The rate at which temperature changes is proportional to the rate at which heat is transferred. The temperature of a sample changes more rapidly if heat is transferred at a high rate and less rapidly if heat is transferred at a low rate. … Answer: the difference in temperature between the two containers of water.What is thermal energy How is it related to temperature?
The thermal energy, or heat, of an object is obtained by adding up the kinetic energy of all the molecules within it. Temperature is the average kinetic energy of the molecules. Absolute zero is the temperature where molecular motion stops and is the lowest possible temperature.
How does temperature affect an object?
Temperature has a direct effect on whether a substance exists as a solid, liquid or gas. Generally, increasing the temperature turns solids into liquids and liquids into gases; reducing it turns gases into liquids and liquids into solids.
How are temperature heat and thermal energy all different?
The core difference is that heat deals with thermal energy, whereas temperature is more concerned with molecular kinetic energy. Heat is the transfer of thermal energy, whereas temperature is a property the object exhibits.
What factors affect thermal energy transfer?
The rate at which an object transfers energy by heating depends on: the surface area, volume and material of the object and the nature of the surface with which the object is in contact. The bigger the temperature difference between a body and its surroundings, the faster the rate at which heat is transferred.Does temperature affect energy?
When the temperature of an object increases, the average kinetic energy of its particles increases. … Therefore, the thermal energy of an object increases as its temperature increases.
How does energy flow depend on temperature?ENERGY, HEAT FLOW, AND LIFE Heat flow moves energy from a higher temperature to a lower temperature. The bigger the difference in temperature between two objects, the faster heat flows between them. When temperatures are the same there is no change in energy due to heat flow.
Article first time published onHow does temperature affect the rate of diffusion?
When temperature increases, the kinetic energy of the particles has increased. The increased motion of the particles causes them to diffuse faster. Therefore, at higher temperatures, the rate at which fluid particles will diffuse is faster than at lower temperatures.
Does warmer or cooler objects have more thermal energy?
Increasing the temperature of something makes these molecules and atoms move around more quickly, giving it more thermal energy. Hot water has more thermal energy than cold water. Colder objects’ molecules and atoms move more slowly and have less thermal energy than warmer objects.
Do hotter things have more energy than colder things?
Artwork: Hotter things have more heat energy than colder things. That’s because the atoms or molecules move around faster in hot things (red, right) than they do in cold things (blue, left). … The more heat you supply, the faster the molecules move and the further apart they get.
Which has more thermal energy Why?
More atoms and higher temperature mean more thermal energy. If all other conditions are the same, substances in gas form have the most thermal energy, followed by liquids, then solids. Temperature can be measured with a thermometer. The matter inside a thermometer expands as its particles gain thermal energy and move.
How does temperature increase?
Greenhouse gases are trapping more heat in the Earth’s atmosphere, which is causing average temperatures to rise all over the world.
What happens to the temperature of an object if it absorbs thermal energy from its surroundings?
Heat, once absorbed as energy, contributes to the overall internal energy of the object. … This means that the particles move more sluggishly and the temperature of the object decreases. The release or absorption of energy in the form heat by an object is often associated with a temperature change of that object.
Is temperature an energy?
Temperature is a measure of the average kinetic energy of the particles in an object.
Does temperature measure energy?
Temperature is a measure of the average kinetic energy of the particles of a substance. The higher the temperature of an object, the higher is its kinetic energy. Kinetic energy is a type of energy associated with motion.
What are three factors affect thermal conductivity?
Factors affect in thermal conductivity of building insulation materials are reviewed. Temperature, moisture content, and density are the most important factors. Other factors include thickness, air velocity, pressing, and aging time. The relationship between main factors with thermal conductivity is presented.
Which temperature will thermal energy naturally flow?
And unless people interfere, thermal energy — or heat — naturally flows in one direction only: from hot toward cold.
Does heat go from hot to cold?
The transfer of heat goes from the hot object to the cold object. … The cold object gets colder and the hot object gets hotter, but energy is conserved.
What is the relationship of the temperature to diffusion?
Temperature: Higher temperatures increase the energy and therefore the movement of the molecules, increasing the rate of diffusion. Lower temperatures decrease the energy of the molecules, thus decreasing the rate of diffusion.
How does temperature affect the rate of reaction?
Increasing the temperature increases the average speed of the reactant molecules. As more molecules move faster, the number of molecules moving fast enough to react increases, which results in faster formation of products.
What is the effect of temperature of temperature on rate of diffusion and kinetic energy?
Explanation: Increasing the temperature increases the kinetic energy of the molecules, which leads them to moving faster and more frequently, and thus increasing the rate of diffusion.
How does the increase in temperature affect the internal energy of a system?
When the temperature of a system increases, the kinetic and potential energies of the atoms and molecules in the system increase. Thus, the internal energy of the system increases, which means that the enthalpy of the system increases – this is true under constant pressure or constant volume.
Does higher temperature mean higher heat?
No it doesn’t. Temperature is the measure of amount of heat energy contained in a substance. Whereas heat capacity is the amount of heat energy required to raise the temperature of unit mass of a substance by 1 degree Celsius at a particular temperature. Heat capacity of a substance varies with temperature.
Which has more thermal energy?
If two objects have the same mass, the object with the higher temperature has greater thermal energy. Temperature affects thermal energy, but temperature isn’t the same thing as thermal energy. That’s because an object’s mass also affects its thermal energy.
What makes something hot or cold thermal energy?
Thermal energy (also called heat energy) is produced when a rise in temperature causes atoms and molecules to move faster and collide with each other. The energy that comes from the temperature of the heated substance is called thermal energy.
What is the relationship between temperature energy and the state of matter?
When thermal energy is added to a substance, its temperature increases, which can change its state from solid to liquid (melting), liquid to gas (vaporization), or solid to gas (sublimation).
What has more thermal energy hot or cold water?
A: The soup is boiling hot and has a temperature of 100 °C, whereas the water in the tub is just comfortably warm, with a temperature of about 38 °C. Although the water in the tub has a much lower temperature, it has greater thermal energy.



