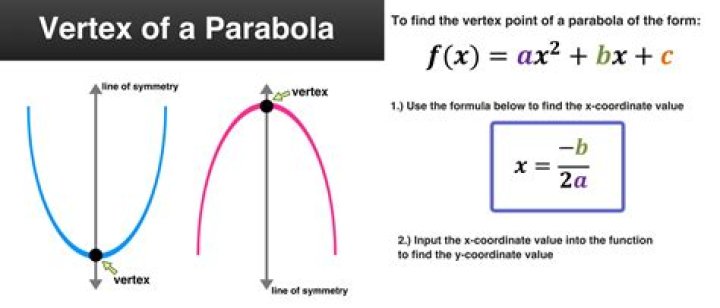
How does lightning fix nitrogen in the atmosphere
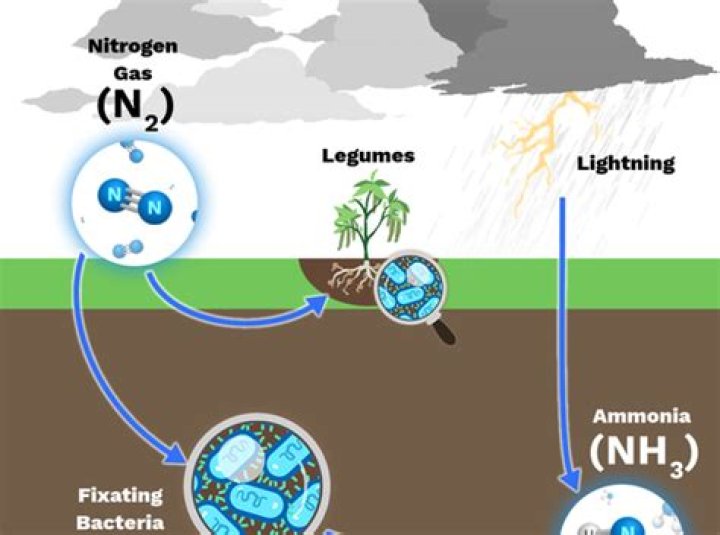
Lightning can also fix nitrogen. The high temperature of a lightning bolt can break the bonds of atmospheric nitrogen molecules. Free nitrogen atoms in the air bond with oxygen in the air to create nitrogen oxides, which dissolve in moisture to form nitrates that are carried to Earth’s surface by precipitation.
How does lightning cause nitrogen fixation?
Each bolt of lightning carries electrical energy that is powerful enough to break the strong bonds of the nitrogen molecule in the atmosphere. Once split, the nitrogen atoms quickly bond to oxygen in the atmosphere, forming nitrogen dioxide.
Does lightning fertilize the ground?
Each lightning bolt carries electrical energy powerful enough to break atmospheric nitrogen bonds. Yes, lightning adds nitrogen to soil, but not directly. … Plants also cannot make use of the nitrogen in the atmosphere so fertilizer is one way to add nitrogen to the soil.
How does nitrogen in the atmosphere get fixed?
Nitrogen gas from the atmosphere is fixed into organic nitrogen by nitrogen-fixing bacteria. This organic nitrogen enters terrestrial food webs. It leaves the food webs as nitrogenous wastes in the soil.Does lightning add nitrogen to rain?
The unbridled energy of lightning shatters the nitrogen molecules in the air. Some of the free nitrogen atoms combine with oxygen to form compounds called nitrates that mix with the rain. … This process is called atmospheric nitrogen fixation, where lightning creates fertilizer in the sky.
How does nitrogen fixation take place during lightning How do plants make use of the nitrates and nitrites present in soil?
Biologically: Nitrogen gas (N2) diffuses into the soil from the atmosphere, and species of bacteria convert this nitrogen to ammonium ions (NH4+), which can be used by plants. … Through lightning: Lightning converts atmospheric nitrogen into ammonia and nitrate (NO3) that enter soil with rainfall.
Does lightning make plants greener?
When it rains, the rain forces the nitrogen to the ground. Microorganisms then take over, converting the nitrogen in the soil and makes grass green. During a thunderstorm, a bolt of lightning can instantly create nitrogen oxide (which is the key ingredient in fertilizers). … Lightning is Mother Nature’s fertilizer!
How does nitrogen fixation work?
Biological nitrogen fixation (BNF) occurs when atmospheric nitrogen is converted to ammonia by an enzyme called nitrogenase. … Many nitrogen-fixing organisms exist only in anaerobic conditions, respiring to draw down oxygen levels, or binding the oxygen with proteins.What causes nitrogen fixation?
Nitrogen is fixed, or combined, in nature as nitric oxide by lightning and ultraviolet rays, but more significant amounts of nitrogen are fixed as ammonia, nitrites, and nitrates by soil microorganisms. … Within the nodules, the bacteria convert free nitrogen to ammonia, which the host plant utilizes for its development.
Which process describes nitrogen fixation?which process describes nitrogen fixation. bacteria take nitrogen from the air and convert in to a form usable by plants. … the process of capture and conservation of nitrogen into a form that is usable by plants.
Article first time published onDoes lightning make nitrogen?
As lightning blasts through the atmosphere, it breaks apart nitrogen molecules. This allows them to combine with oxygen in the air to form nitrogen oxides. The rain dissolves these into nitrates, then carries them to Earth and into the soil.
How does lightning convert nitrogen into nitrates?
With up to a billion volts of electricity, lightning burns at 50,000 degrees, making it hotter than the surface of the sun. When lightning strikes, it tears apart the bond in airborne nitrogen molecules. Those free nitrogen atoms then have the chance to combine with oxygen molecules to form a compound called nitrates.
How much nitrogen is in lightning?
The Story – Greener Grass After Lightning The air contains 78% nitrogen and during lightning some of this is converted to nitrogen dioxide, which dissolves in rain drops, and falls on your lawn. This extra nitrogen works just like a fertilizer; grass absorbs it and becomes greener.
How does nitrogen get into the soil from the atmosphere?
Rain storms contribute atmospheric nitrogen through rain drops that reach the soil. Legumes, such as soybeans, alfalfa and clovers, are plants that can convert atmospheric nitrogen into plant-usable nitrogen.
Why do plants grow better after a thunderstorm?
During a lightning strike, there is enough electrical energy to separate the Nitrogen atoms in the air. Once separated, they fall to the Earth and combine with minerals in the soil to form Nitrates that help plants grow. … Think of lightning as Mother Nature’s fertilizer!
How do plants absorb nitrogen?
Plants absorb nitrogen from the soil in the form of nitrate (NO3−) and ammonium (NH4+). … Nitrate is taken up by several nitrate transporters that use a proton gradient to power the transport. Nitrogen is transported from the root to the shoot via the xylem in the form of nitrate, dissolved ammonia and amino acids.
Does rain add nitrogen to soil?
Because rainwater contains nitrogen in forms that plants can absorb, and plants need nitrogen to grow, farmers have noticed that rainwater stimulates more plant growth than water from other sources. … However, in some cases human activities result in an excess of nitrogen in rainwater.
Why does the grass look so green after it rains?
After it rains, there is more water available in the soil for plants. … When this occurs, a part of the nitrogen is released back into the soil. Ultimately, nitrogen causes growth and helps make things green. As rain falls, nitrogen is pulled out of the atmosphere and in a way this fertilizes the grass.
Why does well watered grass grow better after a thunderstorm?
Although winds and hail can damage a garden, rain during thunder storms can be particularly special for plants. That’s because lightning helps add nitrogen to your garden.
Why are lightning and bacteria important in the nitrogen cycle?
Nitrogen fixation: Nitrogen is required by plants to produce proteins, DNA, RNA, ATP, NAD+, NADP+, Chlorophyll etc. Nitrogen fixation describes the conversion of nitrogen into nitrate, a form that can be used by plants. This is carried out by volcanic action, lightning, industrial activity and by some bacteria.
What are the three different ways of nitrogen fixation?
Plants acquire these forms of “combined” nitrogen by: 1) the addition of ammonia and/or nitrate fertilizer (from the Haber-Bosch process) or manure to soil, 2) the release of these compounds during organic matter decomposition, 3) the conversion of atmospheric nitrogen into the compounds by natural processes, such as …
How does azotobacter fix nitrogen?
Nitrogen Fixing Azotobacter Species as Potential Soil Biological Enhancers for Crop Nutrition and Yield Stability. Biological nitrogen fixation (BNF) refers to a microbial mediated process based upon an enzymatic “Nitrogenase” conversion of atmospheric nitrogen (N2) into ammonium readily absorbable by roots.
How do you fix nitrogen in soil?
- Add Composted Manure.
- Use a Green Manure Crop.
- Plant Nitrogen-Fixing Plants.
- Mix Coffee Grounds in the Soil.
- Use Fish Emulsion.
- Spread Grass Clippings As Mulch.
- Use an Actual Plant Fertilizer.
Can fix atmospheric nitrogen in the soil?
Bacteria such as rhizobium and certain blue-green algae present in the soil can fix atmospheric nitrogen and convert into usable nitrogenous compounds, which are used by plants for the synthesis of plant proteins and other compounds.