How does changing the concentration of enzyme affect the rate of decomposition of h2o
How does changing the concentration of enzyme affect the rate of decomposition of H2O2? Adding more you speed up the decomposition. … Because every enzyme has an optimum and may not do good in other environments.
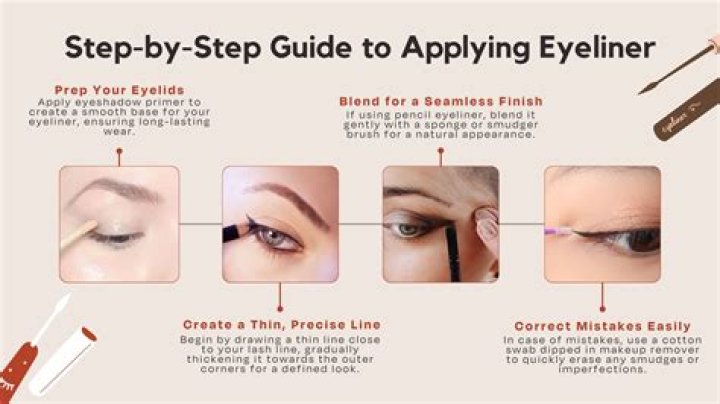
How does changing enzyme concentration affect reaction rate?
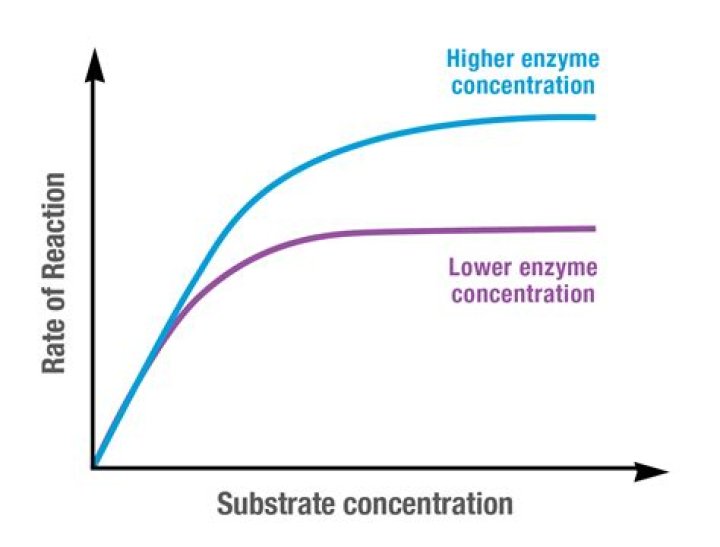
Enzyme concentration: Increasing enzyme concentration will speed up the reaction, as long as there is substrate available to bind to. … Once all of the enzymes have bound, any substrate increase will have no effect on the rate of reaction, as the available enzymes will be saturated and working at their maximum rate.
How does the concentration of catalase affect the rate of decomposition of hydrogen peroxide?
The catalase undergoes spontaneous monomolecular decomposition during the reaction. This inactivation is independent of the concentration of catalase and inversely proportional to the original concentration of peroxide up to 0.4 M. In very high concentrations of peroxide the inactivation rate increases.
How does increasing substrate concentration affect the rate of the enzyme reaction?
Increasing Substrate Concentration increases the rate of reaction. This is because more substrate molecules will be colliding with enzyme molecules, so more product will be formed.Are enzymes affected by concentration of water?
It is found that regardless of the type of reaction, the functionality of enzyme itself is maximum at an optimum level of water, beyond which the enzyme performance is declined due to the loss in enzyme stability.
What happens when enzyme concentration decreases?
The enzyme-bound molecule is called a substrate. Typically, an enzyme is combined with a substrate to reduce the activation energy of a chemical reaction. … This means that as the enzyme concentration decreases, the reaction rate will decrease.
How might differences in enzyme concentrations affect rates of photosynthesis and respiration?
The chemical reactions involved in photosynthesis are controlled by enzymes . … As temperature increases the number of collisions increases, therefore the rate of photosynthesis increases. However, at high temperatures, enzymes are denatured and this will decrease the rate of photosynthesis.
How does changing the concentration of the inhibitor affect the rate of product formation?
Interpretation: As we can see, the rate of product formation increases with increasing substrate concentration. However, in the presence of enzymes the function dp/dt has higher values and a much steeper slope. … Conclusions: The rate of a chemical reaction increases as the substrate concentration increases.How does decreasing the concentration of the substrate affect the enzyme reaction rate?
(B) As the concentration of substrate increases, the enzyme becomes saturated with substrate. As soon as the catalytic site is empty, more substrate is available to bind and undergo reaction. … The rate of reaction when the enzyme is saturated with substrate is the maximum rate of reaction, Vmax.
What is the relationship between substrate concentration and the reaction rate?As the substrate concentration increases the reaction rate does the same, because there is more substrate for the enzyme to react with. This is in fact the linear relationship. As the enzymes become more saturated the reaction levels off.
Article first time published onWhat is the effect of enzyme concentration on catalase activity?
If the substrate concentration is increased then the rate of a catalase reaction will also increase until it reaches the optimal concentration or saturation point and will remain constant. This is because there will be more substrate molecules in a higher concentration therefore a higher frequency of collisions.
How does the concentration of substrate affect the rate of reaction of catalase?
In fact, the catalase reaction is dependent on the substrate concentration. If you have an excess of enzyme but not enough substrate, the reaction will be limited by the substrate availability.
Why does increasing the concentration of h2o2 increase the rate of decomposition?
In many living organisms hydrogen peroxide is a product of metabolism that must be broken down, since in appreciable concentrations it is toxic. The rate of decomposition is increased by the intra-cellular enzyme catalase.
How does water activity affect enzyme activity?
Enzyme activity is influenced by various factors such as aw values, pH and salt content, because higher water can improve enzyme catalyze efficiency while lower water content of sausage can impair the enzyme activity (Monsan and Combes 1984) .
How does enzyme concentration change?
The rate of an enzyme-catalyzed reaction increases with an increase in the concentration of an enzyme. At low temperatures, an increase in temperature increases the rate of an enzyme-catalyzed reaction. At higher temperatures, the protein is denatured, and the rate of the reaction dramatically decreases.
Why are low concentrations of enzymes effective?
In most situations, decreasing enzyme concentration has a direct influence on enzyme activity because each enzyme molecule is able to catalyze only one reaction at a time. … In general, one enzyme binds to one substrate in order to lower the activation energy for one chemical reaction.
Why is enzyme concentration not used to measure the rate of the enzyme catalyzed reaction?
Why is enzyme concentration not used to measure the rate of the enzyme-catalyzed reaction? An enzyme reduces the energy necessary to start a chemical reaction. … hydrogen peroxide is broken down to produce water and oxygen with the help of the enzyme catalase.
How does high heat impact most enzymes?
Increases in temperature increase molecular activity, and can result in a higher rate of collisions between enzymes and substrates. If the temperature rises too high, however, the enzymes could become denatured, and the positive effects of the temperature increase could be nullified.
What factor will increase the rate of photosynthesis?
Temperature, light intensity, carbon dioxide levels, water, oxygen and chlorophyll. The rate of photosynthesis increases as the temperature increases. However, the rate declines after the temperature reaches an optimal level (usually around 35 C). The rate of photosynthesis increases as light intensity increases.
What is the effect of enzyme concentration on enzyme activity explain how enzyme activity changes as enzyme concentration decreases?
We found that the higher the enzyme concentration in the solution, the higher the rate of enzyme activity. As the enzyme concentration decreases, there are fewer enzymes to facilitate chemical reactions.
Does removing heat increase or decrease the rate of reaction?
The reaction rate decreases with a decrease in temperature. … Molecules joined by stronger bonds will have lower reaction rates than will molecules joined by weaker bonds, due to the increased amount of energy required to break the stronger bonds.
How does the concentration of the substrate in an enzyme controlled chemical reaction change over time?
How does the concentration of the substrate in an enzyme-controlled chemical reaction change over time? The substrate concentration will decrease. … Amino acids are linked by peptide bonds with hydrogen bonds between nearby amino and carboxyl groups to give enzymes their two dimensional shape.
Why does the enzyme function vary with substrate concentration?
Before the catalase concentration was saturated with hydrogen peroxide, why did the enzyme function vary with substrate concentration? The enzyme function increased before it was saturated with substrate because it hadn’t yet had enough substrate to saturate all the enzymes.
How does a substrate affect an enzyme quizlet?
The higher the substrate concentration, the faster the reaction. This is because more substrate molecules means a collision between substrate and enzyme is more likely and so more active sites will be used.
How do competitive inhibitors affect the rate of reaction?
Competitive Enzyme Inhibitors work by preventing the formation of Enzyme-Substrate Complexes because they have a similar shape to the substrate molecule. … Therefore less substrate molecules can bind to the enzymes so the reaction rate is decreased.
Does increasing enzyme concentration affect km?
Km is the concentration of substrate at which the enzyme will be running at “half speed”. If you doubled the amount of enzyme, sure the Vmax is going to increase. … The Km is only related with the enzyme,when the enzyme is given,its Km will not change no matter how or what the condition changes.
What is the effect of an inhibitor binding an enzyme quizlet?
inhibitors binds to the active site of the enzyme and “competes” with the substrate for occupation of the site (that type is modeled in the previous slide). the inhibitors binds to the ES complex, but does not bind to free enzyme; thus it may distort the active site and render the enzyme catalytically inactive.
How product concentration affect enzyme activity?
Effect of Product Concentration: The accumulation of reaction products generally decreases the enzyme velocity. For certain’ enzymes, the products combine with the active site of enzyme and form a loose complex and, thus, inhibit the enzyme activity.
What is the effect of substrate concentration on enzyme activity Mcq?
Substrate concentration For a given quantity of enzyme, the rate of reaction increases as the concentration of the substrate is increased.
Which concentration of catalase will produce the fastest reaction rate?
The concentration of catalase that had the fastest reaction time was the 75% catalase concentration, while the slowest reaction time was the 0% catalase concentration.
What feature of catalase most likely is responsible for the change in the rate of conversion of hydrogen peroxide into water and oxygen?
What feature of catalase most likely is responsible for the change in the rate of conversion of hydrogen peroxide into water and oxygen? Catalase speeds up the conversion of hydrogen peroxide into water and oxygen by decreasing the reaction’s activation energy.