How do you sterilize parenteral products
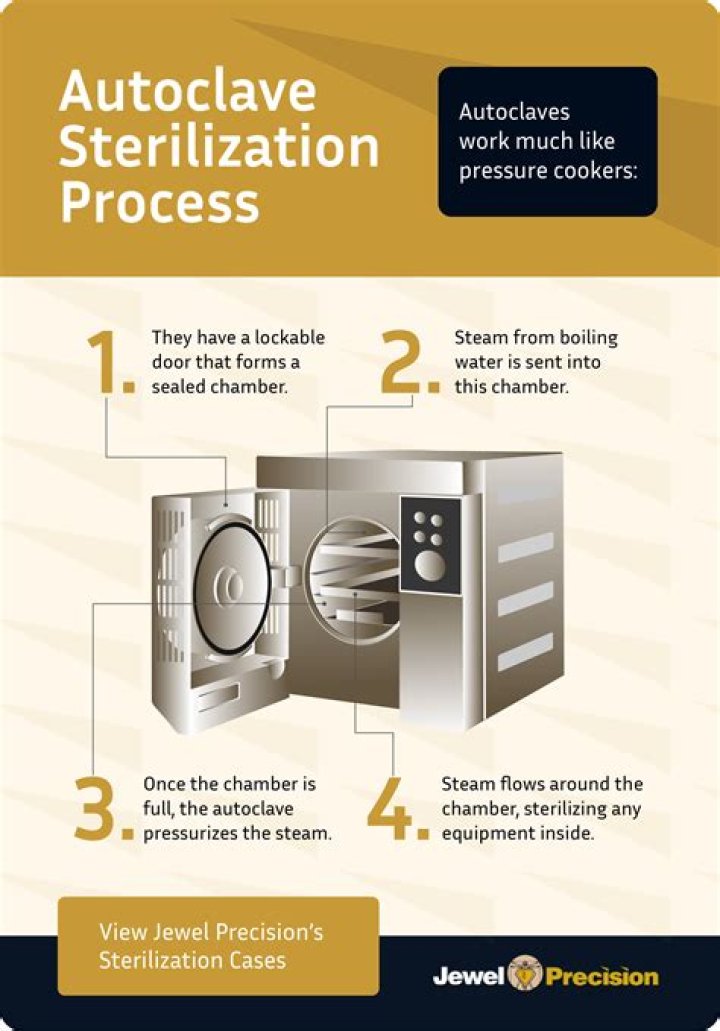
To ensure patient safety, parenteral/injectable drug products must be sterilized to destroy any potential microbial contaminants (fungi, bacteria). The most common sterilization method involves heating under pressure in the presence of water to generate steam; this method is recommended by various pharmacopeias.
What are the 4 methods of sterilization?
- Physical Methods: …
- Radiation Method: …
- Ultrasonic Method: …
- Chemical Method:
How are IV fluids sterilized?
FiltrationFiltration:: Filtration is the preferred method of sterilizingFiltration is the preferred method of sterilizing certain solutions, e.g. IV fluids.
Why autoclave is used in sterilization of parenteral products?
Steam sterilization is carried out in an autoclave, which is an airtight jack-eted chamber designed to maintain a high pressure of saturated hot steam, with the typical temperature of 121°C. … However, steam sterilization cannot be used with materials that are heat sensitive or nonaqueous formulations.What are methods of sterilization?
Sterilization can be achieved by a combination of heat, chemicals, irradiation, high pressure and filtration like steam under pressure, dry heat, ultraviolet radiation, gas vapor sterilants, chlorine dioxide gas etc.
Which sterilization method is suitable for parenteral glassware?
To ensure patient safety, parenteral/injectable drug products must be sterilized to destroy any potential microbial contaminants (fungi, bacteria). The most common sterilization method involves heating under pressure in the presence of water to generate steam; this method is recommended by various pharmacopeias.
How do you sterilize lab equipment?
Wet Heat (Autoclaving) Autoclaving is the most popular method of lab sterilization. This process uses pressurized steam to heat the item that requires sterilization. Autoclaving is an incredibly effective procedure. It will effectively kill all microbes, spores and viruses.
What are the requirements for parenteral preparation?
The parenteral preparations should be free from all types of micro – organisms. An aseptic conditions are required to be maintained during the preparation of Parenteral products and its administration. The parenteral product must pass the test of Sterility. 3 Free from pyrogens.What is parenteral preparation?
Parenteral preparations are defined as solutions, suspensions, emulsions for injection or infusion, powders for injection or infusion, gels for injection and implants. They are sterile preparations intended to be administrated directly into the systemic circulation in human or animal body.
How is terminal sterilization done?Terminal sterilization is achieved by exposure to a physical (e.g., temperature, radiation) or chemical sterilizing agent (e.g., Vaporized Hydrogen Peroxide (VHP), Vaporized Peracetic Acid (VPA), Ethylene Oxide (EO)) for a predetermined extent of treatment.
Article first time published onHow will you sterilize transfusion fluids?
1 The transfusion fluids or intravenous fluids are transferred into washed and dried bottles. These are then properly sealed to exclude and leakage. 2 The bottles are loaded into the sterilizer and its door is closed . 3 The dry saturated steam is admitted into the sterilizer.It will displace the air present in it.
How do terminal Sterilisation and aseptic processes differ?
The aseptic process is more carefully monitored all along production and quality is built into the drug product. Terminal sterilization relies more on the heat treatment procedure carried out at the end of production.
How do you sterilize test tubes?
The best method for sterilizing culture tubes is by autoclaving for 30 minutes at 121 °C (15 psi pressure). Media which solidifies on cooling should be poured out while the tubes are hot.
How do you sterilize serum?
Serum, used to supplement cell culture media, is usually sterilized by filtration because heat-treatment may adversely affect its growth-promoting properties. This filter-sterilization procedure is, however, unreliable because viruses and mycoplasmas may easily pass through the filter.
How are laboratory surfaces sterilized?
Steam Sterilization (Autoclaving) Autoclaving (saturated steam under pressure of approximately 15 psi to achieve a chamber temperature of at least 121oC for a prescribed time) rapidly achieves destruction of microorganisms, decontaminates infectious waste and sterilizes laboratory glassware, media, and reagents.
How do you sterilize glassware?
The most effective and used method of sterilization of glassware is dry heat treatment using hot air oven. On exposing the glassware to dry heat, the inhibition in growth occurs due to denaturation of proteins and oxidation of biomolecules.
How do you sterilize media?
Agar-free media will usually dissolve on gentle agitation. Media containing agar should be heated to dissolve the agar before autoclaving. Bring the medium to the boil without scorching or burning. Most culture media will require final sterilization in an autoclave at 121°C for 20 minutes.
Which method of sterilization is used to sterilize liquid Mcq?
An autoclave is similar to a pressure cooker that uses steam to kill the germs. An autoclave can sterilize solids, liquids, etc.
What is a parenteral solution?
Introduction. Parenteral preparations are defined as solutions, suspensions, emulsions for injection or infusion, powders for injection or infusion, gels for injection and implants. 1. They are sterile preparations intended to be administrated directly into the systemic circulation in humans or animals.
Which parenteral solution is not sterile?
Answer: Parenteral nutrition solutions must be complete, must contain essential and various non-essential amino … This may not be necessary, however, when the central venous catheter is placed under sterile …
In which parenteral preparation preservatives are added?
It was revealed that phenol and benzyl alcohol are the two most common antimicrobial preservatives used in peptide and protein products, while phenoxyethanol is the most frequently used preservative in vaccines.
How are parenteral preparations evaluated?
There are mainly seven quality control tests for parenterals are performed : Leaker test Pyrogen test Particulate test Sterility test Clarity test. Closure integrity test Weight variation test or content uniformity test.
What are the disadvantages of parenteral drug administration?
Disadvantages of parenteral preparations to the patient include lack of drug reversal, risk of infection and emboli, risk of hypersensitivity reactions, and cost.
What is the filtration aid used in filtration of large volume parenteral?
The LVP filtration scheme typically includes a pre-filtration stage using a depth filter to reduce the amount of contaminants and a sterilizing filtration stage using a membrane filter (sometimes a 3-stage cascade scheme including a 0.45 μm membrane filter is used to decrease the bioburden before the sterilizing filter …
What are terminal methods of contraception?
Sterilisation is a terminal method used only for males: Sterilization procedure helps to prevent sperm from meeting with the egg. The method of sterilization varies from male to female. Sterilization in males is termed as vasectomy whereas in females, it is termed as tubectomy.
What are different terminal sterilization process used for ophthalmic?
All large volume parenterals (LVP), small volume parenterals (SVP) and ophthalmic products are terminally sterilized by the moist heat method. Irradiation and ethylene oxide are used for pre-filled syringes and other medical devices. Methods are also useful for the products those are highly heat sensitive.
What is EO sterilization?
Ethylene oxide sterilizers are used to sterilize heat- and moisture- sensitive devices that would be damaged by pure steam or liquid chemical sterilization, including most plastic or rubber products (e.g., catheters, resuscitation bags, anesthesia masks, most fiberoptic instruments), as well as non-heat-sensitive …
How do you sterilize chemicals?
Often chemical sterilizers function by using low temperature, highly reactive gases that come into direct contact with the test article (often through a semi-porous membrane or package.) Liquids – for example, bleach – are also used for sterilization.
How do autoclaves sterilize?
An autoclave is a machine that uses steam under pressure to kill harmful bacteria, viruses, fungi, and spores on items that are placed inside a pressure vessel. The items are heated to an appropriate sterilization temperature for a given amount of time.
What is terminally sterilized medical devices?
Terminal sterilization refers to a sterility assurance level (SAL) of 10− 6 (SAL6 is considered the standard for medical devices [219]) and describes the process that ensures that the medical devices and implants are sterile at the point of use.
What is sterilization and why it is important in plant pathology?
Sterilization refers to any process that removes, kills, or deactivates all forms of life (in particular referring to microorganisms such as fungi, bacteria, spores, unicellular eukaryotic organisms such as Plasmodium, etc.)