How do you calculate the activation energy of a reverse reaction
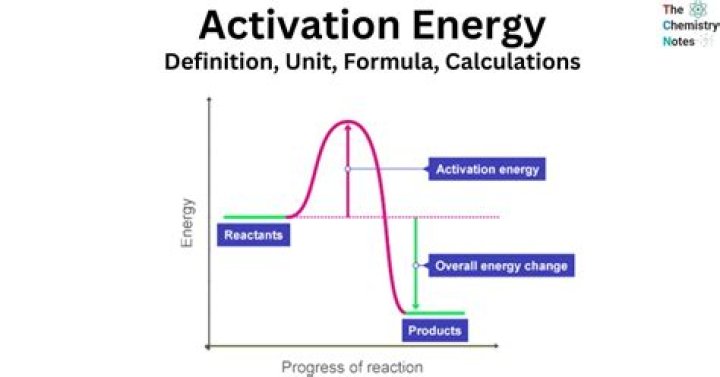
…the activation energy of the reverse reaction is just the difference in energy between the product(s) (right) and the transition state (hill). Thus, for this endothermic reaction, Ea,rev=Ea,fwd−ΔHrxn .
How do you find the reverse activation energy of an exothermic reaction?
It makes sense that the reverse activation energy for an exothermic reaction would be ∆H + Ea since those values added together gives you the height of the hump if you imagine the graph mirrored or going backwards. For an endothermic reaction, the reverse activation energy could be given by Ea – ∆H.
What is the energy change for the reverse reaction?
In other words, for a reversible reaction the energy change is equal for the two reactions, but it has opposite signs. If one reaction is exothermic (energy having a negative value), the opposite reaction is endothermic (energy has a positive value).
What are the activation energies for the forward and reverse reactions?
The activation energies for forward and backward reactions in a chemical reaction are 30.5 and 45.4 kJ mol^-1 respectively.How is the activation energy of the reverse reaction affected by a catalyst?
Catalysts lower the activation energy for the reaction. Catalysts increase the forward rate, while reducing the reverse rate. Catalysts do not alter the energy change between the products and reactants.
Is reverse reaction endothermic or exothermic?
The forward reaction is exothermic (it gives off heat), so the backward reaction is endothermic (takes in heat).
What is the activation energy of the reverse reaction chegg?
The activation energy for the reaction is 7 kJ.
How do you calculate activation energy?
- Step 1: Convert temperatures from degrees Celsius to Kelvin. T = degrees Celsius + 273.15. T1 = 3 + 273.15. …
- Step 2 – Find Ea ln(k2/k1) = Ea/R x (1/T1 – 1/T2) …
- Answer: The activation energy for this reaction is 4.59 x 104 J/mol or 45.9 kJ/mol.
What is backward activation energy?
The activation energy for the backward reaction is equal to the sum of the activation energy of the forward reaction and the enthalpy change of the reaction. Thus the activation energy of the backward reaction is 30+10=40kJmol−1.
How does the energy change for the reverse reaction compared to that for the forward reaction?Energy changes in reversible reactions The same amount of energy is transferred in both the forwards and reverse reaction.
Article first time published onWhat happens in a reversible reaction?
A reversible reaction is a reaction in which the conversion of reactants to products and the conversion of products to reactants occur simultaneously.
How does a catalyst affect the rate of the forward and reverse reactions?
In the presence of a catalyst, both the forward and reverse reaction rates will speed up equally, thereby allowing the system to reach equilibrium faster. … CatalysisA catalyst speeds up a reaction by lowering the activation energy required for the reaction to proceed.
How is the activation energy of a forward reaction affected by the addition of a catalyst?
A catalyst increases the rate of the forward reaction as well as the rate of the backward reaction. … A catalyst causes the decrease in the activation energy for both forward and backward reaction to accelerate them in the specific direction.
What is endothermic reversible reaction?
If a reversible reaction is exothermic (gives out energy) in one direction it is endothermic (takes in energy) in the other direction. … This means that the forward and the backward reactions occur at the same rates.
How do you know if a reaction is forward or reverse?
If K > Q, a reaction will proceed forward, converting reactants into products. If K < Q, the reaction will proceed in the reverse direction, converting products into reactants.
What is a reversible reaction at equilibrium?
A reversible chemical reaction is one in which the products, as soon as they are formed, react to produce the original reactants. At equilibrium, the two opposing reactions go on at equal rates, or velocities, and hence there is no net change in the amounts of substances involved.
What letter represents the activation energy of the reverse reaction?
The letter indicating the activation energy of the reverse reaction is D as it represents the energy of transition state with respect to the…
How do you calculate the enthalpy of activation energy?
The activation energy can be calculated by taking the difference of the transition state enthalpy and the reactant enthalpy. With the enthalpy being defined as the sum of the ground state energy and the statistical mechanically calculated enthalpy.
How do you calculate KN l2 K1?
ln(k2/k1)=(-delta H/R)(1/T2 – 1/T1)
How do you find k?
To determine K for a reaction that is the sum of two or more reactions, add the reactions but multiply the equilibrium constants. The following reactions occur at 1200°C: CO(g)+3H2(g)⇌CH4(g)+H2O(g) K1=9.17×10−2.
How do you calculate activation entropy?
The relationship depends on the molecularity of the reaction: for reactions in solution and unimolecular gas reactions A = (ekBT/h) exp(ΔS‡/R), while for bimolecular gas reactions A = (e2kBT/h) (RT/p) exp(ΔS‡/R).
How does the rate of the reverse reaction compare with the rate of the forward reaction in a reaction at equilibrium?
They are equal, and NONzero. At dynamic chemical equilibrium, the rates of the forward and reverse reactions are equal to each other, i.e. … It is also important to note that the rates of the forward and reverse reactions MUST be nonzero to have a dynamic chemical equilibrium.
What happens to the rate of the reverse reaction as the products are produced?
As reactants are consumed and products are formed, the rate of the forward reaction decreases and the rate of the reverse reaction increases. Eventually, the two rates must become equal.
How do you find the reverse reaction K?
EquationEquilibrium Constant N2(g) + O2(g) NO2(g)Kc = 4.1 x 10-9
Which of the following is a reversible reaction?
Dissolving, evaporation, melting, and freezing are examples of reversible processes.
Which statement is true about reversible reaction?
A reversible chemical reaction can occur in both directions, in that reactants can turn into products and products can turn into reactants.
What is KC formula?
Kc is the equilibrium constant of a chemical reaction. … For the reaction A+B=AB, the equilibrium constant Kc is defined as [AB]/[A][B]. Brackets denote reagent concentrations that must be given in order to compute Kc. As an example, we will calculate Kc for two reactions.
What is a reversible reaction quizlet?
reversible reaction. a reaction which the conversion of reactants into products and the conversion of products into reactants occur simultaneously. chemical equilibrium. a state of balance in which the rates of the forward and reverse reactions are equal.
How does a catalyst decrease activation energy?
A catalyst can lower the activation energy for a reaction by: orienting the reacting particles in such a way that successful collisions are more likely. reacting with the reactants to form an intermediate that requires lower energy to form the product.
Do catalysts forward and backward reactions to the same extent?
A catalyst increases the rate of both the forward and backward reaction to the same extent. Hence, option a is correct. Reaction quotient is the relation between the concentration of reactants and products. Thus the gibbs free energy is unaffected by the addition of catalyst.
When a catalyst is added to a reversible reaction then the value of the equilibrium constant?
A catalyst does not influences the values of equilibrium constant.