How do molecular chaperones work
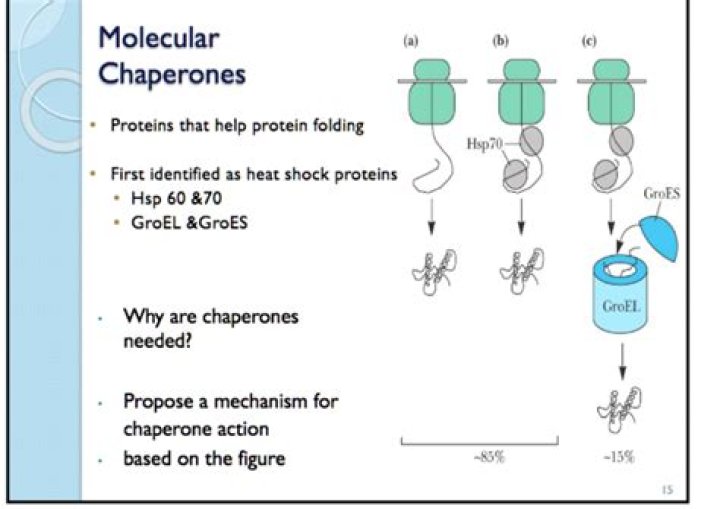
Molecular chaperones interact with unfolded or partially folded protein subunits, e.g. nascent chains emerging from the ribosome, or extended chains being translocated across subcellular membranes. They stabilize non-native conformation and facilitate correct folding of protein subunits.
How do chaperone proteins work?
Chaperones are proteins that guide proteins along the proper pathways for folding. They protect proteins when they are in the process of folding, shielding them from other proteins that might bind and hinder the process. … Heat, in general, destabilizes proteins and makes misfolding more common.
What is the function of the molecular chaperone?
Molecular chaperones, including Hsp60s, Hsp70s, Hsp90s and sHsps, assist in the folding of unfolded and misfolded polypeptides by stabilization of folding intermediates and prevention of protein misfolding and aggregation. Several chaperones also function to reactivate aggregated proteins.
How do molecular chaperones assist proteins to fold?
Chaperones prevent aggregation and incorrect folding by binding to and stabilizing partially or totally unfolded protein polypeptides until the polypeptide chain is fully synthesized. They also ensure the stability of unfolded polypeptide chains as they are transported into the subcellular organelles.Where are molecular chaperones found?
Chaperonins are characterized by a stacked double-ring structure and are found in prokaryotes, in the cytosol of eukaryotes, and in mitochondria. Other types of chaperones are involved in transport across membranes, for example membranes of the mitochondria and endoplasmic reticulum (ER) in eukaryotes.
What is the role of chaperones in the major protein degradation pathways?
During the pathogenesis, molecular chaperones play a central role in the refolding, degradation, and disaggregation of these pathogenic protein species. Extensive studies have shown that molecular chaperones promote the removal of pathogenic misfolded proteins and their aggregates.
Do molecular chaperones require ATP?
During and after protein translation, molecular chaperones require ATP hydrolysis to favor the native folding of their substrates and, under stress, to avoid aggregation and revert misfolding.
What statement best describes how chaperones perform their function?
Practice: Which statement best describes how chaperones perform their function? They prevent intermolecular hydrophobic interactions to facilitate proper protein folding.Why are molecular chaperones critical for protein folding?
Molecular chaperones are diverse families of multidomain proteins that have evolved to assist nascent proteins to reach their native fold, protect subunits from heat shock during the assembly of complexes, prevent protein aggregation or mediate targeted unfolding and disassembly.
Are molecular chaperones enzymes?Molecular chaperones as enzymes that catalytically unfold misfolded polypeptides.
Article first time published onHow many molecular chaperones are there?
Current structural information divides the chaperones into five major classes based on their observed molecular weights: Hsp60, Hsp70, Hsp90, Hsp104, and the small Hsps. Aside from their differences in size, the structures of these different classes are quite divergent.
What is the nature of molecular chaperones?
Molecular chaperones interact with unfolded or partially folded protein subunits, e.g. nascent chains emerging from the ribosome, or extended chains being translocated across subcellular membranes. They stabilize non-native conformation and facilitate correct folding of protein subunits.
How the molecular chaperones plays role in the modification of the nascent proteins?
Rather, chaperones catalyze protein folding by assisting the self-assembly process. They appear to function by binding to and stabilizing unfolded or partially folded polypeptides that are intermediates along the pathway leading to the final correctly folded state.
Are chaperones catalysts?
Rather, chaperones are ubiquitous unfolding catalysts that can ubiquitously recognize abnormal (hydrophobic) structures on the surface of their misfolded and aggregated polypeptide substrates and thus serve as effective passive fences and active cellular defenses against proteotoxic conformers causing protein …
What are molecular chaperones quizlet?
Molecular Chaperones. auxiliary protein that protect and stabilize folding proteins. Protein Folding. protein structure assumes its functional state or conformation from random coil.
Why is BiP considered integral to the role fulfilled by the ER?
BiP, an HSP70 molecular chaperone located in the lumen of the endoplasmic reticulum (ER), binds newly-synthesized proteins as they are translocated into the ER and maintains them in a state competent for subsequent folding and oligomerization.
Do chaperone proteins degrade proteins?
Conversely, chaperone-assisted degradation of proteins that are structurally perturbed, but still functional, has also been linked to disease, as in cystic fibrosis [8]. At present, our understanding of what determines whether a chaperone commits to a folding or a degradation mode is limited.
Can misfolded proteins be fixed?
CONN: The pharmacoperone interacts physically with the molecule and creates the shape that passes through the cell’s quality control system and because of that, even misfolded proteins can be refolded and trafficked correctly in the cell, thereby restoring them to function.
What's the difference between chaperones and Chaperonins?
The key difference between chaperons and chaperonins is that the chaperones perform a wide array of functions including folding and degradation of the protein, aiding in protein assembly, etc., whereas the key function of chaperonins is to assist in the folding of large protein molecules.
Are lectins chaperones?
N-glycosylation allows newly synthesized glycoproteins to interact with a lectin-based chaperone system in the endoplasmic reticulum.
What do molecular chaperones do in post translational modification?
Post-translational modifications (PTMs) encompass a number of specific chemical modifications that occur on proteins following their synthesis. … Molecular chaperones are proteins involved in the folding or unfolding of other proteins as well as assembly or disassembly of larger macromolecular complexes.
How do proteins fold and unfold?
Folded proteins are held together by various molecular interactions. During translation, each protein is synthesized as a linear chain of amino acids or a random coil which does not have a stable 3D structure. The amino acids in the chain eventually interact with each other to form a well-defined, folded protein.