How can electrons move
When electric voltage is applied, an electric field within the metal triggers the movement of the electrons, making them shift from one end to another end of the conductor. Electrons will move toward the positive side.
How do electrons keep moving?
An electron does not “rotate” around its nucleus. Rather, it stays in the vicinity of the nucleus because of the attractive force between its negative charge and the nucleus’ positive charge.
Why can only electrons move?
Explanation: Electrons are located in shells surrounding the nucleus of the atom,whereas protons are present inside the nucleus. Electrons can move out of the shells on providing energy, but movement of proton will require a very high amount of energy (so as to break the nuclear force).
What forces electrons to move?
EMF is electromotive force. EMF causes the electrons to move in a particular direction. EMF is measured in units called volts.Can electrons stop moving?
No, it’s not possible to stop an electron. because of the simple fact, it has to obey the Heisenberg uncertainty relation with respect to place and momentum. In the extreme case (theoretically) we can measure the electron’s momentum with absolute certainty.
Why do electrons flow to earth?
The ground is an attractive place for electricity to flow because it is positively charged, only more so when the tiny particles in the atmosphere collide, filling clouds with negatively charged particles. (These are also called ions.)
Do electrons move up or down?
In larger and larger atoms, electrons can be found at higher and higher energy levels (e.g. 3s and 3p). … Almost immediately the excited electron gives up the extra energy it holds, usually in the form of light, and falls back down to the lower energy level again. Florescence is a phenomenon of moving electrons.
Can electrons be transferred?
Whenever electrons are transferred between objects, neutral matter becomes charged. For example, when atoms lose or gain electrons they become charged particles called ions. Three ways electrons can be transferred are conduction, friction, and polarization. In each case, the total charge remains the same.Why do electrons flow in the first place?
Electrons are charged and hence they flow in the first place.
How fast do electrons move?Drift velocity, the average speed at which electrons travel in a conductor when subjected to an electric field, is about 1mm per second. It’s the electromagnetic wave rippling through the electrons that propagates at close to the speed of light.
Article first time published onCan an electron be still?
Since you can’t have both, an exact position and an exact velocity (zero or not) you can not force a particle to stand still. This is an excellent question, more at the end.
Why do atoms move?
Atoms move by either electrical force or force of gravity. Even gravity requires electric force, because otherwise, except in extreme cases, Gravity is too weak to attract a single isolated atom. Electric forces arise because of: ions which induce an electric field.
Do electrons move in waves?
Electrons in higher-energy atomic states vibrate more quickly. Because an electron is a quantum object with wave-like properties, it must always be vibrating at some frequency. … Furthermore, an electron in a stable atomic state does not move in the sense of waving through space.
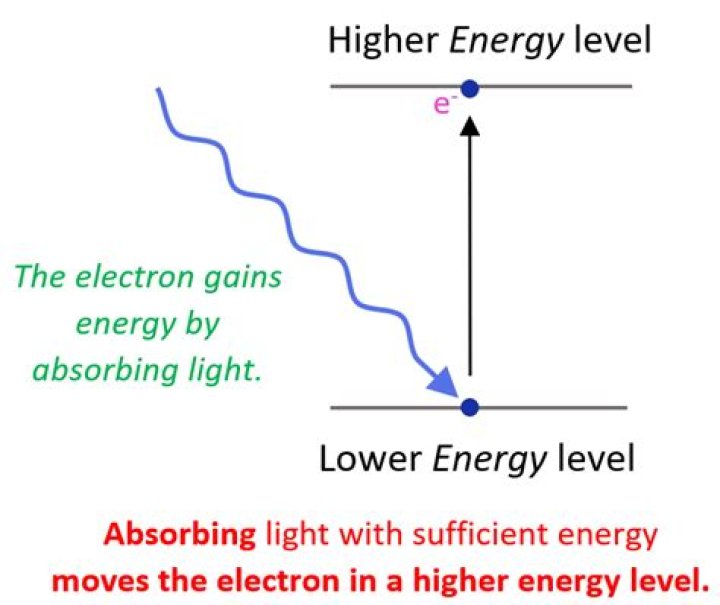
How can an electron move from one energy level to another?
To move from one energy level to another, an electron must gain or lose just the right amount of energy. … When atoms absorb energy, electrons move into higher energy levels. These electrons lose energy by emitting light when they return to lower energy levels.
Why do electrons move and not protons?
Electrons move freely within the structure of an atom but protons are bound in the nucleus and therefore immobile. Conductivity will therefore occur when electrons move from one atom to another and not protons due to their immobility.
Is Earth ocean of electrons?
Earth can provide extra electrons to neutralise the positive charge or absorb electrons to neutralise the negative charge. Thus, Earth is known as a huge ocean of charges. … It is electrically neutral and contains equal amounts of positive and negative charges.
Can the earth become charged?
The Earth surface is negatively charged. According to the charge-neutrality principle, the electric charge of the whole Earth is ZERO. The reason why the surface of the Earth is negatively charged remains to be clarified.
Where do electrons go in an atom?
Electrons are found in different levels — or orbitals — surrounding the nucleus. The electrons can be found at any point in their orbital. The orbitals can be shaped as a sphere, as lobes — which kind of look like two squashes put together at the small ends — or in the shape of a doughnut around the nucleus.
What is electron shift?
However, when we study in depth about charge transfer we will understand that the reaction is only partial. Therefore, it can be described as electron shift rather than electron transfer or electron loss where complete loss and gain of electrons occurs in Hydrogen and oxygen, respectively.
What is a transfer of electrons called?
The attraction between oppositely charged ions is called an ionic bond, and it is one of the main types of chemical bonds in chemistry. Ionic bonds are caused by electrons transferring from one atom to another.
What is electron transfer in physics?
Electron transfer is a fundamental process which plays a central role in physics, chemistry, and biology. Biological electron transfer reactions are required for respiration, photosynthesis, and redox reactions of intermediary metabolism.
Can electron move with speed of light?
No, electrons have mass and nothing with mass can travel at the speed of light.
Do electrons move faster than light?
Electrons have rest mass, so they cannot travel as fast as light. Although they be nearly equal to the speed of light but cannot be equal to the speed of light.
Does electron really exist?
According to Dirac, at any point in space, the electron neither exists nor doesn’t exist. It can only be described as a mathematical function. The same is true for the quarks that make up the atom’s nucleus, as they too are fermions, which behave according to the Dirac equation.
Can an electron be in 2 places at once?
About 80 years ago, scientists discovered that it is possible to be in two locations at the same time — at least for an atom or a subatomic particle, such as an electron. For such tiny objects, the world is governed by a madhouse set of physical laws known as quantum mechanics.
Why do particles vibrate?
The particles are held together too strongly to allow movement from place to place but the particles do vibrate about their position in the structure. With an increase in temperature, the particles gain kinetic energy and vibrate faster and more strongly.
Can atoms be moved?
Scientists using the latest microscopic technology have found they can move individual atoms around a surface at will, a breakthrough that could have profound implications in the ultra-small world of electronics and even in industry. … The capability to move atoms around freely might provide an answer to this problem.
Which atoms move around the most?
The atoms and molecules in gases are much more spread out than in solids or liquids. They vibrate and move freely at high speeds. A gas will fill any container, but if the container is not sealed, the gas will escape.
Is air a atom?
The Air We Breathe. Air is actually a collection of very small molecules; oxygen gas and nitrogen gas are molecules made from two atoms each. Only the noble gases readily exist as single atoms.
Are electrons a wave or particle?
Along with all other quantum objects, an electron is partly a wave and partly a particle. To be more accurate, an electron is neither literally a traditional wave nor a traditional particle, but is instead a quantized fluctuating probability wavefunction.
How are the electrons excited?
When an electron temporarily occupies an energy state greater than its ground state, it is in an excited state. An electron can become excited if it is given extra energy, such as if it absorbs a photon, or packet of light, or collides with a nearby atom or particle.