Does heat affect water hardness?
Does heat affect water hardness?
As you can se boiling the water causes the precipitation of solid calcium carbonate or solid magnesium carbonate. This removes the calcium ions or magnesium ions from the water, and so removes the hardness. Generally an increase in water temperature causes an increase in the solubility of most salts.
What causes softening of hard water?
Water softening is achieved either by adding chemicals that form insoluble precipitates or by ion exchange. On a small scale, chemicals used for softening include ammonia, borax, calcium hydroxide (slaked lime), or trisodium phosphate, usually in conjunction with sodium carbonate (soda ash).
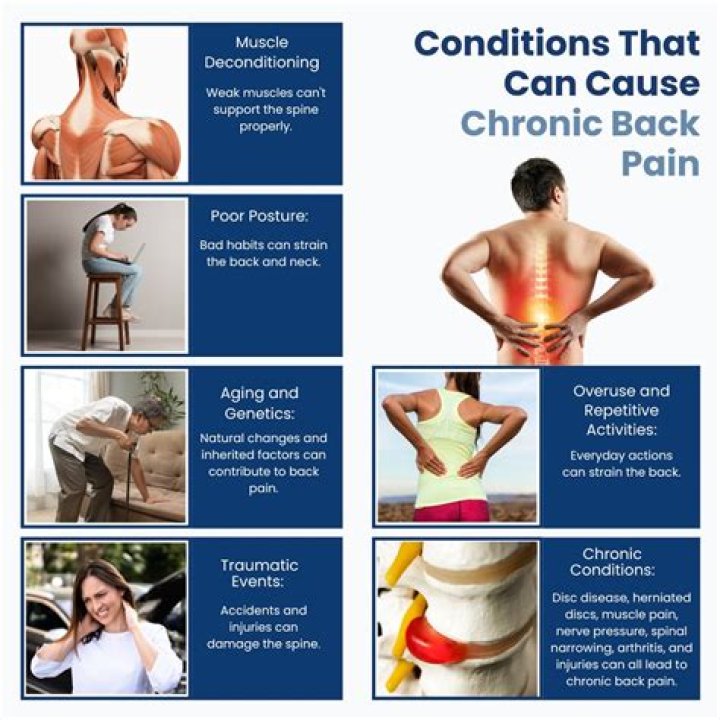
What is main cause of water hardness?
Water becomes hard by being in contact with soluble, divalent, metallic cations. The two main cations that cause water hardness are calcium (Ca2+) and magnesium (Mg2+). Calcium is dissolved in water as it passes over and through limestone deposits.
What if I only soften the hot water?
Soften the hot water only Use less water and salt by only softening the hot water and leaving the cold water hard. You probably won’t even notice the difference and it will reduce your salt and wastewater by nearly half. It won’t harm your water heater or plumbing.
What is temperature of normal water?
What is the number of a normal temperature of water? – Quora. room temperature of water is 20-25 degree Celsius,Freezing point 0 Celsius,Boining point 100 Celsius. On average, the temperature of tap water is roughly 13 degrees Celsius.
How is softening of hard water done?
Water is softened when calcium and magnesium—the hardness ions—are collected by tiny resin beads through a process called ion exchange. When hard water comes in contact with the resin beads, the calcium and magnesium are pulled out of the water and replaced with sodium or potassium, leaving your water soft.
What is the main disadvantage of hard water?
Explanation: Hard water consumes more amount of soap. Hard water requires more scaling in boilers. Hard water also corrodes and incrusts pipes.
What temperature of water is healthy?
When drinking hot beverages, research recommends an optimal temperature of between 130 and 160°F (54 and 71°C). Temperatures above this can cause burns or scalds. For an extra health boost and some vitamin C, try adding a twist of lemon to hot water to make lemon water.
What causes hard water and soft water to be hard?
Based on the hardness, we have two main types of water; hard water and soft water. As mentioned above, the principal causes of water hardness are calcium and magnesium minerals.
Why does hard water have calcium in it?
Hard water contains dissolved magnesium ions and calcium ions, which can get into the water when it comes into contact with limestone and other rocks that contain calcium compounds. This can happen, for example, when rainwater flows over rocks on its way to a reservoir.
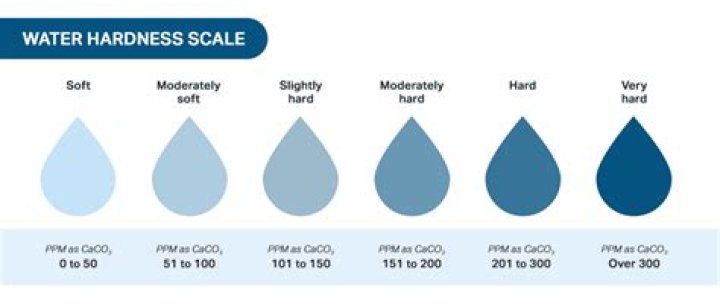
Which is the best definition of hard water?
The simple definition of water hardness is the amount of dissolved calcium and magnesium in the water. Hard water is high in dissolved minerals, largely calcium and magnesium. You may have felt the effects of hard water, literally, the last time you washed your hands.
What do you need to know about the hardness of water?
When using hard water, more soap or detergent is needed to get things clean, be it your hands, hair, or your laundry. One of the most common causes of cloudy dishes and glassware is hard water.
What’s the difference between hard and soft water?
Soft water is the opposite of hard water, which means you have less calcium in the water, whereas hard water has a lot of calcium. But is soft water bad for hot tubs?
Why is too hard water bad for hot tubs?
Water that is too hard (too high in calcium) can cause scale deposits to build up in your pipes and equipment. It can also lead to cloudy water. Also, make sure you keep your pH and alkalinity balanced as that affects how the calcium affects your water. Use a water softening system to reduce the calcium levels in your hard water.
When using hard water, more soap or detergent is needed to get things clean, be it your hands, hair, or your laundry. One of the most common causes of cloudy dishes and glassware is hard water.
The simple definition of water hardness is the amount of dissolved calcium and magnesium in the water. Hard water is high in dissolved minerals, largely calcium and magnesium. You may have felt the effects of hard water, literally, the last time you washed your hands.