Does Argon form covalent bonds
The extremely stable noble gasses
Does argon make covalent or ionic bonds?
These elements—helium, neon, argon, krypton, xenon, and radon—do not form compounds very easily, which suggests that they are especially stable as lone atoms. What else do the noble gas elements have in common? Except for helium, they all have eight valence electrons.
What element forms covalent bonds?
PropertyIonicCovalentHow bond is madeTransfer of e-Sharing of e-Bond is betweenMetals and nonmetalsNonmetals
What type of bond does argon form?
Molecules containing argon can be van der Waals molecules held together very weakly by London dispersion forces. Ionic molecules can be bound by charge induced dipole interactions. With gold atoms there can be some covalent interaction.Why argon does not form either ionic bonds or covalent bonds?
Argon belongs to inert group elements which already has a complete octet electronic configuration and hence they are stable. As Argon is already stable, there is no need of losing and gaining electrons and hence there is no formation of ionic bonds.
Does argon have lattice?
solid, liquid or gas. Solid Argon crystallizes in a “face-centered cubic” (fcc) lattice.
Can argon form compounds?
Under standard conditions argon is an odorless and colorless gas. It is also an inert gas, meaning that it typically doesn’t react with other elements to form compounds. … Argon has been found to form one neutral compound with fluorine and hydrogen called argon fluorohydride (HArF).
Does argon form an ion with a charge?
Argon can donate one electron to become a +1 ion, or it can accept one electron to become a -1…Can argon bond with lithium?
Argon is a noble gas. Meaning, it does not simply “bond” with anything. These special gasses have a VERY strong resistance to joining with other atoms. So, to answer your question, no, you can not bond Argon and Lithium under any circumstance.
Is argon a compound or element?argon (Ar), chemical element, inert gas of Group 18 (noble gases) of the periodic table, terrestrially the most abundant and industrially the most frequently used of the noble gases.
Article first time published onWhat elements do not form covalent bonds?
The elements that is unlikely for form covalent bond are K and Ar.
What are the examples of covalent bond?
- Hydrogen (H2) Hydrogen (H) is the simplest of all elements. …
- Oxygen (O2) The valency of oxygen (O) is two, which means that it requires two electrons to complete its outermost (valence) shell. …
- Nitrogen (N2) …
- Water (H2O) …
- Carbon Dioxide (CO2) …
- Methane (CH4) …
- Ammonia (NH3) …
- Carbon Monoxide (CO)
What structure is argon?
Atomic Number18Shell structure (Electrons per energy level)[2, 8, 8]Electron Configuration[Ne] 3s2 3p6Valence Electrons3s2 3p6Oxidation State
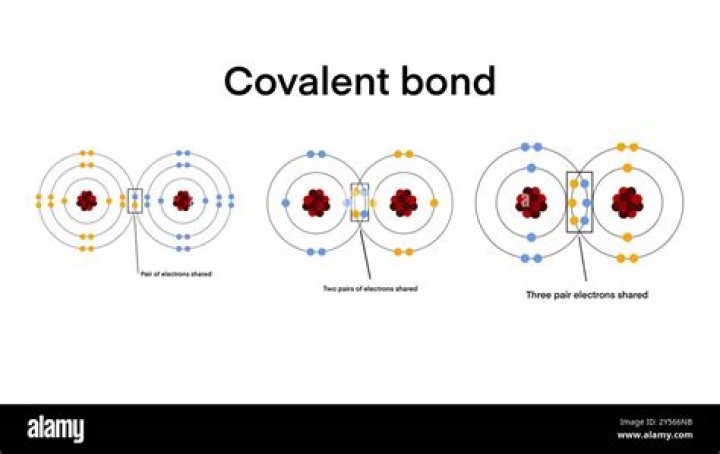
How chemical bonds are formed?
Bonds form when atoms share or transfer valence electrons. Valence electrons are the electrons in the outer energy level of an atom that may be involved in chemical interactions. Valence electrons are the basis of all chemical bonds.
Which of the following is a covalent compound?
(d) CCl4 is a covalent compound. CCl4 is a covalent compound as the atoms of carbon and chlorine are characterized by four bonds of non-polar covalent.
Why argon does not form an oxide compound?
All noble gases have full s and p outer electron shells (except helium, which has no p sublevel), and so do not form chemical compounds easily. Because of their high ionization energy and almost zero electron affinity, they were not expected to be reactive.
What element reacts with argon?
Argon is a noble gas and it does not react with any other element. It does not even react at high temperatures or under any other special conditions. One succeeded in producing only one argon compound that was very instable, under extremely low temperatures.
Why does neon and argon not form compounds?
Noble gas atoms are located in column 18 of the periodic table. The valence shells of these atoms are already completely filled. Therefore, they are already stable and do not need to form compounds in order to fill their valence shells.
What does argon do in the atmosphere?
Argon provides an inert atmosphere in which welded metals will not oxidise. Argon is a colourless, odourless gas that is totally inert to other substances. Argon is often used when an inert atmosphere is needed. It is used in this way for the production of titanium and other reactive elements.
What types of atoms are in argon?
chlorine – argon Ne Ar Kr Full tableGeneralName, Symbol, Numberargon, Ar, 18Chemical seriesnoble gasesGroup, Period, Block18 (VIIIA), 3, p
Is argon a discrete atom or molecule?
The 2 noble gases are of course mono-atomic; some may be quibble with the distinction, but while these are discrete gaseous particles, they are atomic, not molecular. So, xenon, and argon exist as discrete atoms that exist in the gas phase.
Is argon made of atoms or molecules?
The element argon has always been a loner. It’s one of the inert gases that normally exist as single atoms. But in the 23 August issue of Nature, chemists report that they persuaded argon to mingle a little and form a compound with other elements.
Does argon form negative ions?
Positively and negatively charged ions are observed for an argon/oxygen gas mixture, resulting from gas phase collisions and from interaction with the sputtering target. Major differences are noticed to the pure argon case.
Does argon gain or lose electrons?
The noble gases are unreactive, because their outer electron shells are full. A full shell of outer electrons is a particularly stable arrangement. This means that noble gas atoms neither gain nor lose electrons easily; they react with other atoms with great difficulty, or not at all.
Is argon a renewable resource?
ArgonPronunciation/ˈɑːrɡɒn/ (AR-gon)Appearancecolorless gas exhibiting a lilac/violet glow when placed in an electric field
How is argon formed?
On Earth, the vast majority of argon is the isotope argon-40, which arises from the radioactive decay of potassium-40, according to Chemicool. But in space, argon is made in stars, when a two hydrogen nuclei, or alpha-particles, fuse with silicon-32. The result is the isotope argon-36.
Which of these Cannot form covalent bond Why?
Element with atomic no 17 cannot form covalent bond easily because for the formation of covalent bond it must share 7 electrons which is highly impossible.
How do you identify a covalent bond?
There is a couple different ways to determine if a bond is ionic or covalent. By definition, an ionic bond is between a metal and a nonmetal, and a covalent bond is between 2 nonmetals. So you usually just look at the periodic table and determine whether your compound is made of a metal/nonmetal or is just 2 nonmetals.
What are 3 types of covalent bonds?
- Single bonds occur when two electrons are shared and are composed of one sigma bond between the two atoms.
- Double bonds occur when four electrons are shared between the two atoms and consist of one sigma bond and one pi bond.
Does H2O have covalent bonds?
Water (H2O), like hydrogen fluoride (HF), is a polar covalent molecule. When you look at a diagram of water (see Fig. 3-2), you can see that the two hydrogen atoms are not evenly distributed around the oxygen atom.
Why argon gas is used in spectrometer?
The Reasons of Using Argon Gas Isolate the atmosphere by flushing the surface so that the spectrometer won’t detect the elements in the atmosphere. Spectrometer can detect the element, which has its characteristic peak below the wavelength 200nm correctly. In the composition of alloy, the element Argon is not ignored.